Introduction
Carbon-14 radiolabelling is one of the most important techniques in modern pharmaceutical and biochemical research. For more than half a century, this isotope has allowed scientists to trace the behaviour of complex organic molecules in living systems with remarkable sensitivity. By incorporating a radioactive carbon atom into a drug candidate or biochemical substrate, researchers can track the compound through metabolic pathways, quantify its distribution in biological tissues, and determine its elimination from the body.
Although carbon-14 radiolabelling is widely used, the physical process that makes it possible is often discussed only briefly. The key to the method lies in the emission of beta particles during radioactive decay. These emitted electrons provide the measurable signal that allows scientists to detect minute quantities of labelled molecules in biological samples.
At the same time, the emission of beta particles can occasionally influence the stability of the labelled compound itself. In certain molecules, particularly those containing reactive functional groups or weak chemical bonds, ionising events from beta particles may trigger radiolysis, leading to gradual chemical degradation.
This article explores the role of beta particles in carbon-14 radiolabelling, the mechanisms by which they enable detection, and the circumstances under which beta radiation can cause molecular instability. Particular attention is given to classes of compounds that may be susceptible to radiolytic processes, including organic azides, peroxides, diazo compounds, nitroaromatic molecules, and halogenated organic structures.
Understanding these processes is important not only for chemists synthesising radiolabelled molecules, but also for pharmacologists, radiochemists, and regulatory scientists working in drug development.
Carbon-14 as a Radiotracer
Carbon-14 is a naturally occurring radioactive isotope of carbon that contains six protons and eight neutrons in its nucleus. The isotope is unstable and undergoes radioactive decay with a half-life of approximately 5730 years. Because carbon is present in nearly all organic molecules, carbon-14 can be incorporated into a wide variety of chemical structures without significantly altering their chemical properties.
This property makes carbon-14 extremely useful as a radiotracer. When a drug molecule is synthesised with a carbon-14 atom in a specific position, it behaves chemically like the non-radioactive compound. However, the radioactive atom allows the molecule to be tracked using sensitive detection techniques.
Carbon-14 radiolabelling is used extensively in:
- drug metabolism studies
- pharmacokinetic investigations
- environmental fate studies
- biochemical pathway research
- human mass balance studies
These investigations are frequently required during pharmaceutical development to determine how a new drug behaves in the body.
Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency expect radiolabelled studies to be conducted for many drug candidates. The information obtained from these studies helps determine metabolic pathways, identify circulating metabolites, and assess whether potentially toxic products are formed during drug metabolism.
The Physics of Carbon-14 Beta Decay
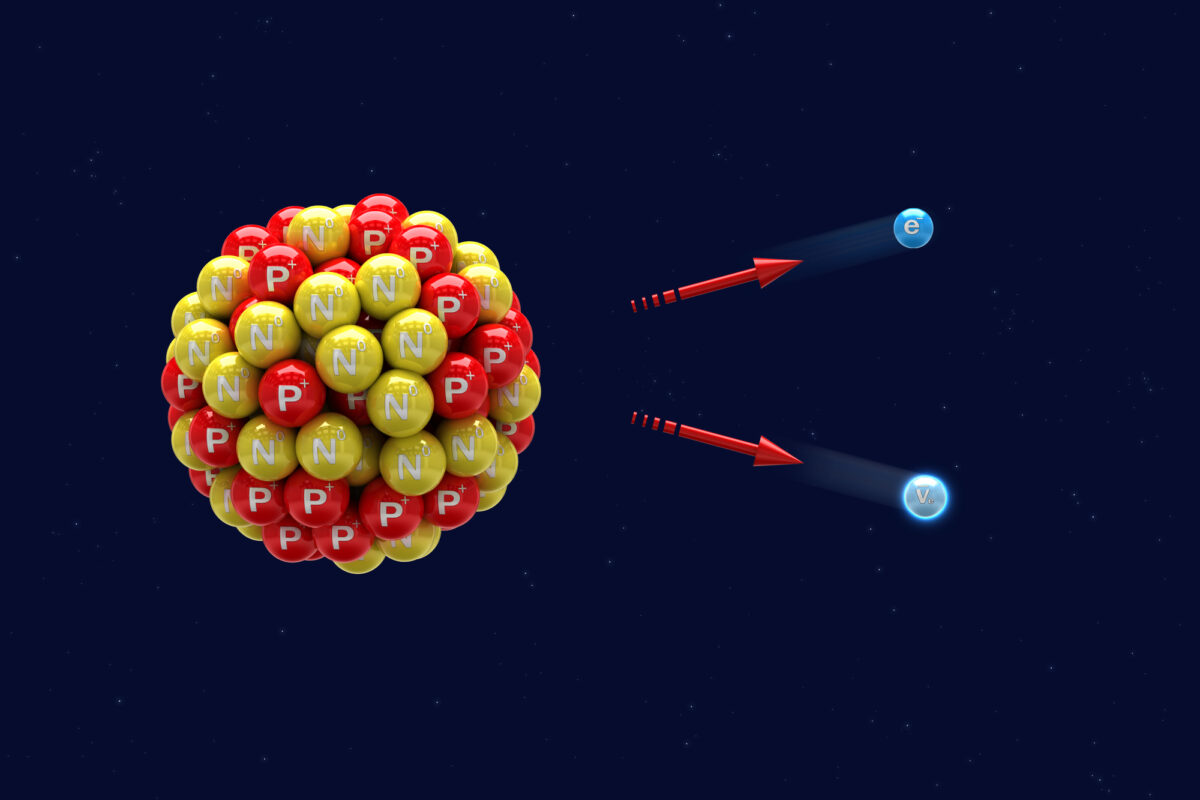
The usefulness of carbon-14 as a tracer arises from its mode of radioactive decay. Carbon-14 undergoes beta minus decay, a nuclear process in which a neutron within the nucleus converts into a proton.
During this transformation, three particles are produced:
- a beta particle, which is a high-energy electron
- an antineutrino
- a new atomic nucleus
The nuclear reaction can be represented as:
¹⁴C → ¹⁴N + β⁻ + ν̅
As a result of this process, the carbon-14 atom becomes nitrogen-14.
The emitted electron carries kinetic energy and travels through the surrounding material until it loses energy through interactions with nearby molecules. These interactions cause ionisation and excitation events that form the basis of radiochemical detection methods.
Carbon-14 emits relatively low-energy beta particles, with a maximum energy of approximately 156 keV. This energy is significantly lower than that of many other beta-emitting isotopes used in nuclear science. The limited energy of beta particles means they travel only short distances before losing their energy.
Typical penetration distances are:
- a few millimetres in air
- fractions of a millimetre in biological tissue
- extremely short distances in dense materials
Because the particles do not travel far, carbon-14 radiation is relatively easy to shield and poses minimal external hazard in laboratory environments.
Detection of Carbon-14 Through Beta Emissions
The emitted beta particles provide the measurable signal used to detect carbon-14 labelled molecules. When beta particles interact with surrounding matter, they transfer energy to electrons in nearby atoms, causing excitation or ionisation.
Several analytical techniques exploit these interactions.
Liquid Scintillation Counting
The most common detection method is liquid scintillation counting. In this technique, a sample containing a carbon-14 labelled compound is mixed with a scintillation cocktail. The cocktail contains molecules that emit flashes of light when excited by ionising radiation.
When a beta particle passes through the solution, it excites the scintillator molecules, which then release small pulses of light. Sensitive detectors, such as photomultiplier tubes, measure these light pulses and convert them into electrical signals. The number of detected pulses corresponds to the amount of radioactivity present in the sample.
Liquid scintillation counting allows extremely sensitive measurement of carbon-14, often detecting picogram quantities of labelled compounds.
Radio-HPLC
Carbon-14-labelled compounds are frequently analysed by radio-HPLC. In this approach, the components of a mixture are separated by high-performance liquid chromatography, and a radioactivity detector monitors the eluting fractions.
The beta particles emitted by the labelled compound generate scintillation events in the detector, producing signals corresponding to individual metabolites.
This technique is widely used in metabolite profiling studies.
Accelerator Mass Spectrometry
Another powerful analytical technique is accelerator mass spectrometry (AMS). AMS measures the number of carbon-14 atoms directly rather than detecting their radioactive decay. The method is extremely sensitive and allows detection of carbon-14 at levels far below those measurable by conventional radiometric methods.
AMS has enabled the development of microtracer studies, in which extremely small doses of radiolabelled drugs can be administered to human volunteers.
Radiolysis in Carbon-14 Labelled Compounds
Although the beta particles emitted by carbon-14 are relatively low in energy, they can still produce localised ionisation events in molecules containing the radioactive atom. When the decay event occurs within a labelled compound, the emitted electron may interact with neighbouring atoms, generating radicals or excited states.
In many molecules, these interactions have little effect on chemical stability. However, compounds containing weak bonds or energy-rich functional groups may be more susceptible to degradation.
This process is known as radiolysis, a phenomenon in which radiation induces chemical reactions that break or rearrange molecular bonds.
Radiolysis can occur through several mechanisms:
- direct ionisation of the molecule
- formation of radical intermediates
- interaction with solvent-derived radicals
- secondary reactions involving oxygen or water
Because carbon-14 has a very long half-life, the rate of decay is extremely slow. For most compounds used in laboratory studies, radiolysis is minimal. Nevertheless, when radiolabelled molecules are stored for long periods or contain particularly reactive functional groups, radiolytic degradation may become noticeable.
Organic Azides and Radiation Sensitivity
Organic azides represent one class of compounds that can be sensitive to radiolytic processes. The azide functional group consists of a linear arrangement of three nitrogen atoms attached to an organic substituent.
The N–N bonds within the azide group contain considerable stored energy. When energy is supplied by heat, ultraviolet light, or ionising radiation, the azide may decompose, releasing nitrogen gas and generating a reactive intermediate, a nitrene.
Nitrenes are highly reactive species capable of inserting into carbon–hydrogen bonds, forming new chemical structures or triggering rearrangement reactions.
In a carbon-14 labelled azide, the beta particle emitted during radioactive decay may produce local ionisation that initiates cleavage of the azide group. Although the probability of such an event is low, it becomes more significant when compounds are stored for extended periods.
Examples of azide-containing molecules that could be susceptible to this type of degradation include benzyl azides, aryl azides, and alkyl azides containing carbon-14 labels.
Because azides are already chemically reactive, radiolysis may accelerate their decomposition or lead to the formation of secondary products.
Peroxides and Hydroperoxides
Another class of molecules susceptible to radiation-induced degradation includes organic peroxides and hydroperoxides. These compounds contain an oxygen–oxygen bond, which is considerably weaker than most carbon–carbon or carbon–oxygen bonds.
When exposed to energy, the O–O bond may undergo homolytic cleavage, producing radical species such as alkoxy radicals or hydroxyl radicals.
In carbon-14-labelled peroxides, the beta particle emitted during radioactive decay may provide sufficient energy to initiate this cleavage. The resulting radicals can then react with neighbouring molecules, leading to chain reactions that degrade the compound.
This behaviour is particularly relevant in biological systems, where lipid hydroperoxides and other oxidised molecules may be studied using radiolabelled tracers.
Diazo Compounds and Carbene Formation
Diazo compounds represent another group of molecules that contain high-energy nitrogen-based functional groups. These compounds typically contain the –N₂ group attached to a carbon atom.
When energy is supplied through heat, light, or radiation, diazo compounds can release nitrogen gas and generate highly reactive carbene intermediates.
Carbenes can insert into chemical bonds, form cyclopropanes, rearrange molecular frameworks, or react with nearby functional groups.
In a carbon-14-labelled diazo compound, beta-particle emission could trigger decomposition by providing the energy required to release nitrogen. Although this type of reaction is uncommon under normal storage conditions, radiation may accelerate the degradation of sensitive molecules.
Because diazo compounds are already reactive species, radiolysis represents an additional pathway for decomposition.
Nitroaromatic Compounds
Nitroaromatic molecules contain strongly electron-withdrawing nitro groups attached to aromatic rings. These groups can participate in redox reactions involving electron transfer.
Radiation can produce solvated electrons and other reactive species in solution. When such species interact with nitro groups, they may form radical anions, which can subsequently convert to nitroso or hydroxylamine intermediates.
Carbon-14-labelled nitroaromatic compounds may therefore undergo slow chemical changes when exposed to radiation over long periods.
Examples of molecules that may exhibit this behaviour include nitrobenzene derivatives and nitroimidazole compounds used in biochemical research.
Halogenated Organic Compounds
Halogenated organic molecules containing chlorine or bromine atoms can also be susceptible to radiation-induced reactions. Carbon–halogen bonds may undergo homolytic cleavage when exposed to energetic electrons.
In the presence of radiation, radical intermediates may form, leading to dehalogenation or substitution reactions.
Carbon-14 labelled halogenated molecules used in environmental or biochemical studies may therefore show evidence of slow radiolytic degradation if stored for extended periods.
Examples include halogenated hydrocarbons and chlorinated organic acids.
Factors That Influence Radiolysis
Several factors influence the likelihood of radiolytic degradation in carbon-14 labelled compounds.
One important factor is specific activity, which refers to the amount of radioactivity present per unit mass of compound. Molecules with higher specific activity undergo more decay events within a given period, increasing the probability of radiation-induced reactions.
Another factor is storage time. Because carbon-14 decays slowly, radiolysis usually becomes noticeable only after prolonged storage.
The chemical structure of the compound also plays a crucial role. Molecules containing weak bonds or reactive functional groups are more likely to undergo radiolytic decomposition.
Environmental conditions may also influence stability. The presence of oxygen, water, or reactive solvents can generate additional radical species, which can contribute to degradation.
Temperature can further affect radiolysis by increasing the rate of secondary chemical reactions.
Strategies for Minimising Radiolysis
To maintain the stability of radiolabelled compounds, several practical strategies are commonly used.
Radiolabelled molecules are often stored at low temperatures, which reduces the rate of chemical reactions that may follow radiation events.
Samples may also be stored under inert atmospheres, such as nitrogen or argon, to minimise reactions involving oxygen.
Solvents such as ethanol or acetonitrile are frequently used because they can stabilise reactive intermediates and reduce the likelihood of radical chain reactions.
In some cases, radical scavengers or stabilising agents may be added to prevent secondary reactions.
These precautions help ensure that radiolabelled compounds remain chemically stable throughout the duration of experimental studies.
Self-Radiolysis Within Labelled Molecules
An interesting aspect of carbon-14 chemistry is the possibility of self-radiolysis, in which the decay event occurs within the molecule itself. Because the radioactive atom is part of the molecular structure, the emitted beta particle begins its trajectory inside the compound.
This localised energy deposition may lead to ionisation events in the immediate vicinity of the labelled carbon atom. In most cases, the energy is dissipated without significant chemical consequences. However, in particularly sensitive molecules, the energy transfer may initiate bond cleavage or radical formation.
Self-radiolysis is therefore a phenomenon that chemists consider when designing radiolabelled compounds, particularly those intended for long-term storage.
Carbon-14 Radiolabelling in Modern Drug Development
Even with the potential for radiolysis in certain chemical structures, carbon-14 remains one of the most valuable tools in pharmaceutical science. Radiolabelled compounds allow researchers to study drug metabolism with extraordinary sensitivity and precision.
In human mass balance studies, for example, volunteers may receive a small dose of a carbon-14 labelled drug. Researchers then measure the radioactivity in biological samples, such as plasma, urine, and faeces, to determine how the drug is absorbed, distributed, metabolised, and eliminated.
These studies provide essential information about metabolic pathways and help identify potentially toxic metabolites.
Advances in analytical techniques, particularly accelerator mass spectrometry, have further expanded the capabilities of carbon-14 radiolabelling. Microtracer studies using extremely small doses of radiolabelled compounds are now possible, reducing radiation exposure while still providing valuable pharmacokinetic information.
Conclusion
Carbon-14 radiolabelling remains an indispensable technique in chemical and pharmaceutical research. The isotope’s long half-life and ability to integrate seamlessly into organic molecules make it an ideal tracer for studying metabolic processes and drug distribution.
The method relies on the emission of beta particles during radioactive decay. These electrons interact with surrounding materials to produce detectable signals that allow scientists to measure extremely small quantities of radiolabelled compounds.
While the energy of carbon-14 beta particles is relatively low, the ionisation events they produce can occasionally trigger radiolytic reactions in chemically sensitive molecules. Compounds containing reactive functional groups, including azides, peroxides, diazo structures, nitroaromatics, and halogenated molecules, may be particularly susceptible under certain conditions.
Understanding these processes allows chemists to design more stable radiolabelled compounds and adopt appropriate storage strategies to minimise degradation.
Even so, the advantages of carbon-14 radiolabelling far outweigh its limitations. The technique continues to provide critical insights into drug metabolism, biochemical pathways, and environmental chemistry. As analytical technologies continue to advance, carbon-14 will likely remain a central tool in tracing the complex journeys of molecules through biological systems.
Disclaimer
The information presented in this article is intended for educational and informational purposes only. While every effort has been made to ensure accuracy, the content should not be interpreted as professional, regulatory, or safety guidance. Handling radioactive materials, including carbon-14 radiolabelled compounds, requires appropriate training, regulatory approval, and adherence to institutional safety procedures. Readers should consult qualified professionals and relevant regulatory authorities before conducting any work involving radioactive substances.
home »