OPEN MEDSCIENCE REVIEW | August 10, 2025
Abstract: This mini-review provides a concise yet detailed examination of carbon-14 (¹⁴C) radiolabelling, covering its principles, methods, and applications across scientific disciplines. It begins with the discovery, physical properties, and production of ¹⁴C, before outlining key radiolabelling concepts such as specific activity, label positioning, and stability. Synthetic approaches using [¹⁴C]CO₂, [¹⁴C]cyanide, and [¹⁴C]methyl iodide are discussed alongside purification and protection strategies. Analytical techniques, including liquid scintillation counting, radio-HPLC, and accelerator mass spectrometry, are presented for assessing purity and activity. Applications in pharmaceuticals, environmental studies, and biochemistry are illustrated through selected case studies. The review also summarises safety practices, regulatory frameworks, and ethical considerations, and highlights emerging trends such as late-stage labelling, flow radiochemistry, and computational design tools. This synthesis of theoretical and practical perspectives offers a compact reference for researchers employing ¹⁴C radiolabelling in modern science.
Keywords: carbon-14 radiolabelling, radioisotope tracer studies, radiochemistry techniques, drug metabolism (ADME), environmental fate analysis, accelerator mass spectrometry (AMS).
1 – Introduction to Carbon-14 Radiolabelling
Carbon-14, often denoted as ¹⁴C, is one of the most widely used radioactive isotopes in research involving tracer methodologies. Its unique combination of a long half-life, chemical identity with stable carbon isotopes, and the ability to be incorporated into a wide variety of organic molecules makes it a versatile and powerful tool in both fundamental and applied sciences.
Radiocarbon was first identified in 1940 by Martin Kamen and Sam Ruben at the University of California, Berkeley, during studies of nuclear reactions involving deuterons and graphite. Shortly thereafter, its utility in studying metabolic pathways became apparent, particularly through the work of Melvin Calvin and colleagues, who employed carbon-14-labelled compounds to map the photosynthetic carbon fixation pathway—work that ultimately earned Calvin the Nobel Prize in Chemistry in 1961.
The value of carbon-14 radiolabelling lies in its ability to function as a tracer because carbon atoms form the backbone of all organic molecules; substitution of a ¹²C atom for a ¹⁴C atom in a compound generally does not alter the compound’s chemical or biological behaviour. This allows the labelled compound to participate in biochemical processes without affecting the kinetics or thermodynamics of the system under study. The radioactive decay of ¹⁴C to nitrogen-14, with emission of a low-energy beta particle, can then be detected with high sensitivity using liquid scintillation counting, autoradiography, or other radiometric techniques.
Radiolabelling with carbon-14 is especially prominent in the pharmaceutical industry, where it is used to investigate the absorption, distribution, metabolism, and excretion (ADME) profiles of new drug candidates. By tracking the fate of a labelled drug in animal models and humans, researchers can obtain a complete mass balance and identify metabolic products, even when these are present at trace levels. Similar strategies are applied in environmental science to study the degradation of pesticides, pollutants, and other organic chemicals, often under regulatory guidelines.
The scope of carbon-14 radiolabelling is not limited to biological systems. In chemistry, radiolabelled substrates have been used to investigate reaction mechanisms and to determine kinetic isotope effects. In geoscience, the naturally occurring ¹⁴C isotope forms the basis for radiocarbon dating, a method widely used to estimate the age of archaeological samples and geological formations.
In this review, the focus will be on the principles, methodologies, and applications of carbon-14 radiolabelling as used in controlled laboratory and industrial settings. Each section will address a specific aspect of the topic, beginning with the fundamental properties of the isotope, moving through the design and synthesis of labelled compounds, and concluding with safety considerations, regulatory issues, and future developments in the field.
2 – Fundamentals of Carbon-14
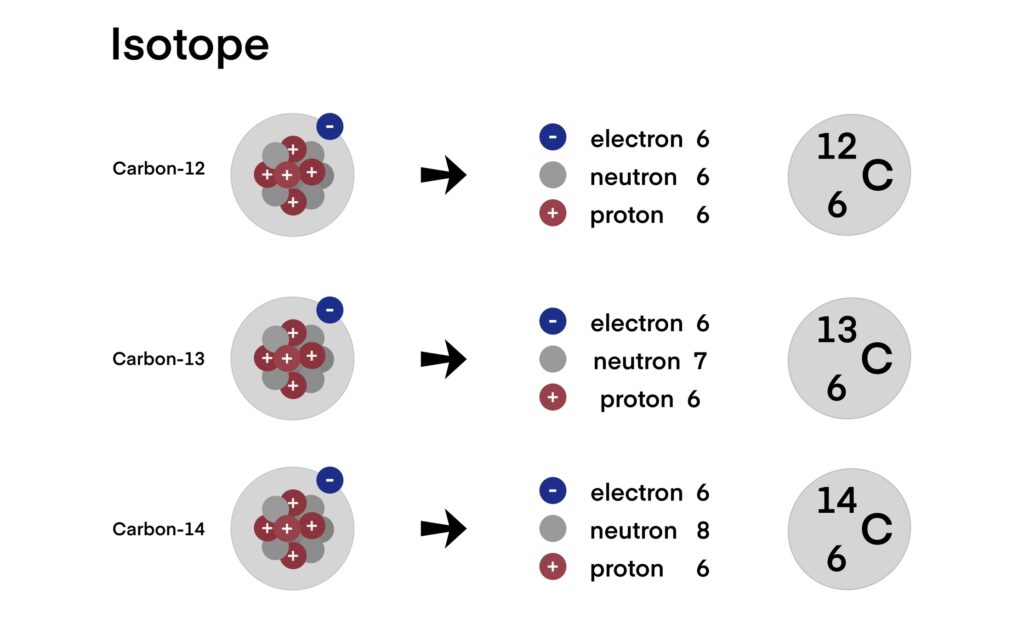
2.1 Physical and Nuclear Properties
Carbon-14 (¹⁴C) is a radioactive isotope of carbon with an atomic number of 6 and a mass number of 14. It contains eight neutrons and six protons in its nucleus. The isotope is unstable, undergoing β⁻ decay to nitrogen-14 (¹⁴N) with the emission of a low-energy beta particle and an antineutrino. The decay process can be represented as:
14C → 14N + β− + ν‾e
The half-life of carbon-14 is approximately 5,730 years, which makes it a long-lived radionuclide. This extended half-life is one of the reasons it is so valuable for radiolabelling, as it allows experiments and long-term studies without significant decay loss over the timescale of weeks, months, or even years.
The β⁻ particle emitted during decay has a maximum energy of about 156 keV and an average energy of roughly 49 keV. This low-emission energy results in relatively low penetration in matter, typically only a few millimetres in tissue or a few centimetres in air, making external radiation hazards minimal. However, internal exposure through ingestion, inhalation, or skin absorption of contaminated materials poses significant health risks.
2.2 Production of Carbon-14
In nature, ¹⁴C is continuously formed in the upper atmosphere through the interaction of cosmic rays with atmospheric nitrogen. High-energy neutrons from cosmic radiation collide with ¹⁴N atoms, producing ¹⁴C via the reaction:
14N + n → 14C + p
The newly formed ¹⁴C rapidly oxidises to carbon dioxide, mixes with atmospheric CO₂, and is incorporated into plants through photosynthesis, subsequently entering the food chain.
For laboratory and industrial purposes, naturally occurring carbon-14 is insufficient in both quantity and purity. Artificial production is achieved mainly in nuclear reactors by neutron irradiation of nitrogen-containing targets, such as aluminium nitride or graphite doped with nitrogen. This controlled production allows the generation of ¹⁴C-labelled starting materials such as barium carbonate, sodium carbonate, or labelled methane, which serve as precursors for chemical synthesis.
2.3 Radioactive Decay and Detection
The β⁻ decay of ¹⁴C is purely electronic in nature, with no accompanying gamma radiation. This simplifies shielding requirements, but it also means that detection relies on techniques optimised for beta particles. The most common detection methods are:
- Liquid Scintillation Counting (LSC) – The sample is mixed with a scintillation cocktail, where beta particles excite the scintillant, producing photons that are detected by photomultiplier tubes. This method is highly sensitive and can measure activity down to a few disintegrations per minute (dpm).
- Gas-Flow Proportional Counting – Suitable for dry or solid samples, this technique detects ionisation in a gas chamber caused by beta particles. It is often used for radiocarbon dating rather than synthetic chemistry applications.
- Autoradiography – Used for visualising the distribution of radioactivity in thin samples, such as chromatographic plates or biological tissue sections, by exposing them to X-ray film or phosphor screens.
2.4 Safety Considerations and Regulatory Frameworks
Although ¹⁴C is a low-energy beta emitter, it is classified as a radioactive material under national and international regulations. The primary hazard is internal contamination. The committed effective dose coefficient for ingestion of ¹⁴C (in organic form) by an adult is approximately 5.8×10−10Sv/Bq, which underscores the need for strict handling protocols.
Work with ¹⁴C in a laboratory setting typically requires:
- Operation in designated radiochemistry areas with controlled access.
- Use of personal protective equipment (PPE) such as lab coats, gloves, and safety glasses.
- Contamination monitoring using Geiger-Müller probes (for high-activity sources) and swipe tests analysed by LSC.
- Appropriate storage and disposal of radioactive waste in compliance with regulations from agencies such as the International Atomic Energy Agency (IAEA), the International Commission on Radiological Protection (ICRP), and relevant national bodies.
Key Points: The long half-life, low radiation emissions, and high detectability of carbon-14 make it especially suitable for tracer research in diverse scientific areas. Understanding its nuclear properties, production routes, and detection methods is essential before embarking on synthetic radiolabelling work. The next section will build on this foundation by examining the principles of radiolabelling—how to design experiments that make optimal use of ¹⁴C without compromising the molecule’s chemical or biological function.
3 – Principles of Radiolabelling
3.1 Concept of Isotopic Labelling and Tracer Methodology
Radiolabelling is the process of incorporating a radioactive isotope, such as carbon-14, into a molecule so that the molecule’s fate can be tracked through chemical, biological, or environmental systems. The central principle is that isotopic substitution with ¹⁴C does not alter the compound’s chemical identity or behaviour, as the isotope behaves identically to the stable isotopes ¹²C and ¹³C in chemical bonding and reactivity.
This chemical indistinguishability is what makes carbon-14 an ideal tracer. Once introduced into a compound at a defined position, the isotope’s radioactive decay can be detected with high sensitivity, even at very low concentrations, allowing precise quantification of distribution, transformation, and persistence.
3.2 Specific Activity and Molar Activity
Specific activity refers to the radioactivity per unit mass of a labelled compound, usually expressed in becquerels per milligram (Bq/mg) or curies per millimole (Ci/mmol). It is determined by the proportion of carbon atoms in the molecule that are ¹⁴C.
Molar activity is related but normalised per mole of compound, allowing direct comparison between molecules of different sizes.
Two key factors influence specific activity:
- Isotopic abundance – whether the label is introduced at a single carbon position or at multiple sites.
- Purity of the label – whether there is dilution by unlabelled carbon during synthesis.
High specific activity is often desirable for detecting low-concentration species in biological systems. However, for certain mechanistic or environmental studies, lower specific activities may suffice and can be more cost-effective.
3.3 Choice of Labelling Position
The position at which ¹⁴C is introduced can greatly affect the utility of the labelled compound:
- Metabolically stable positions are chosen when the aim is to track the entire molecule’s fate, such as in total mass balance studies of pharmaceuticals.
- Metabolically labile positions can be labelled if the objective is to study specific cleavage pathways or intermediate formation.
- Symmetrical molecules may require labelling in a way that avoids isotopic scrambling during synthesis or metabolism.
Selecting the labelling site also requires consideration of synthetic accessibility. For example, introducing ¹⁴C into the carbonyl carbon of a carboxylic acid may be straightforward using [¹⁴C]carbon dioxide, whereas introducing the isotope into an aromatic ring may require multi-step synthesis.
3.4 Minimising Isotopic Exchange and Degradation
Once incorporated, the ¹⁴C label must remain in place during the course of the experiment. Isotopic exchange with unlabelled carbon sources or degradation of the labelled functional group can compromise data integrity.
Common causes of label loss include:
- Decarboxylation of labelled carboxylic acids under thermal or acidic conditions.
- Isotope dilution during synthetic steps involving large excesses of unlabelled reagents.
- Transcarbonylation or transmethylation reactions in vivo or in vitro.
Strategies to mitigate these issues involve:
- Choosing metabolically resistant positions.
- Using mild reaction conditions during synthesis.
- Protecting labelled groups with stable protecting groups until final deprotection.
3.5 Purity and Stability Considerations
After synthesis, it is essential to confirm the radiochemical purity—the proportion of radioactivity associated with the desired compound. Impurities can arise from incomplete reactions, side-products, or degradation. Analytical methods such as radio-HPLC or radio-TLC, coupled with UV or MS detection, are used to verify both chemical and radiochemical purity.
Radiolabelled compounds should be stored under conditions that minimise radiolysis (self-decomposition due to radiation). For ¹⁴C-labelled organics, this typically means low temperatures (often -20 °C or below), exclusion of light, and sometimes storage under an inert atmosphere to prevent oxidation.
Key Points: Radiolabelling with carbon-14 is both a science and an art. Achieving the desired label position, specific activity, and stability requires careful planning and understanding of both chemistry and the biological or environmental context of the study. The next section will focus on synthetic methods—how ¹⁴C is introduced into molecules using precursor compounds and functional group transformations.
4 – Synthetic Methods for Carbon-14 Labelling
4.1 Overview of Synthetic Planning for Radiolabelling
Synthetic routes for introducing carbon-14 into organic molecules require careful consideration of:
- The labelled precursor to be used.
- The position of the label within the target structure.
- The scale of synthesis (milligram to gram quantities).
- Minimising isotope loss during reaction and purification.
Because ¹⁴C is expensive and radioactive, radiolabelling syntheses are typically carried out at the smallest practical scale, often using micro-scale or semi-preparative methods, while ensuring that the labelled compound meets purity and specific activity requirements.
4.2 Common Carbon-14 Precursors
Commercial suppliers provide a variety of ¹⁴C-labelled small molecules that serve as starting points for incorporation into more complex targets. Common examples include:
- [¹⁴C]Carbon dioxide (¹⁴CO₂)
- Generated from barium [¹⁴C]carbonate or supplied in sealed ampoules.
- Useful for direct carboxylation reactions.
- Often employed in the synthesis of labelled carboxylic acids, esters, and amides.
- [¹⁴C]Barium carbonate (Ba[¹⁴C]CO₃)
- Solid, stable, and easy to store.
- Converted to ¹⁴CO₂ by acid treatment.
- [¹⁴C]Potassium or sodium cyanide (K[¹⁴C]CN / Na[¹⁴C]CN)
- Versatile precursor for nitrile synthesis and subsequent functional group interconversion.
- Requires stringent safety protocols due to combined radiological and chemical toxicity.
- [¹⁴C]Methyl iodide ([¹⁴C]CH₃I)
- Used for methylation of heteroatoms (N, O, S) and carbon atoms.
- Typically prepared from [¹⁴C]methanol.
- [¹⁴C]Methanol
- Applied in O-methylation and as a building block for larger structures.
- [¹⁴C]Urea, [¹⁴C]formaldehyde, and other small intermediates
- Useful in specific biochemical or agrochemical contexts.
4.3 Functional Group Transformations for Incorporating ¹⁴C
4.3.1 Carboxylation with [¹⁴C]CO₂
Carboxylation reactions are a cornerstone of ¹⁴C radiolabelling, especially when introducing the isotope into the terminal carbon of a chain. Examples include:
- Grignard reagents + [¹⁴C]CO₂ → labelled carboxylic acids.
- Organolithium reagents + [¹⁴C]CO₂ for sensitive molecules.
- Transition-metal-catalysed carboxylation of aryl halides.
4.3.2 Nitrile Chemistry via [¹⁴C]Cyanide
Nitriles prepared from K[¹⁴C]CN can be converted into:
- Amides (via hydration).
- Carboxylic acids (via hydrolysis).
- Amines (via reduction).
This flexibility makes cyanide one of the most important building blocks for ¹⁴C incorporation.
4.3.3 Alkylation with [¹⁴C]Methyl Iodide
Methylation allows the introduction of the isotope into O-methyl, N-methyl, or S-methyl groups. This is particularly useful for labelled drugs or metabolites where methylation patterns are significant in metabolism studies.
4.3.4 Carbonylation Reactions
Using labelled carbon monoxide ([¹⁴C]CO) or derivatives, transition-metal-catalysed carbonylations can introduce the isotope into aldehydes, ketones, esters, and amides. This approach is useful for aromatic carbonyl compounds.
4.4 Multi-Step Labelling Strategies
For complex targets, direct incorporation of ¹⁴C into the final molecule is often impractical. Instead, the isotope is introduced into a key intermediate that can be elaborated to the final structure.
Example:
- [¹⁴C]CO₂ → labelled benzoic acid
- Benzoic acid → labelled benzoyl chloride
- Benzoyl chloride → labelled benzamide
- Benzamide → further derivatisation to target drug candidate.
4.5 Use of Protecting Groups
Protecting groups can shield reactive functional groups during multi-step synthesis, preventing side reactions and isotopic loss. For example:
- Esterification of labelled acids to methyl esters for stability during synthesis, followed by hydrolysis to release the acid.
- N-protection of amines during radiolabelling steps involving electrophiles.
4.6 Purification and Isolation
Given the high expense of ¹⁴C materials, purification processes should be designed to minimise losses. Common techniques include:
- Preparative thin-layer chromatography (pTLC) with radio-TLC monitoring.
- Semi-preparative high-performance liquid chromatography (HPLC) equipped with a radioactivity detector.
- Crystallisation from minimal volumes of solvent.
Key Points: Synthetic radiolabelling with carbon-14 relies on a small but versatile toolkit of labelled precursors, combined with careful functional group transformations and protection strategies. The next section will focus on analytical techniques, covering both qualitative and quantitative methods to verify the identity, purity, and specific activity of labelled compounds.
5 – Analytical Techniques
5.1 Introduction
Once a ¹⁴C-labelled compound has been synthesised, verifying its chemical identity, radiochemical purity, and specific activity is essential before it can be used in experimental work. The sensitivity of ¹⁴C detection allows for precise quantification even at picogram levels, but it also demands robust analytical methods to ensure the reliability of results.
The analytical strategy typically combines separation techniques to isolate the compound of interest and detection methods to measure its radioactivity. These approaches can be used for both quality control in synthesis and monitoring during biological or environmental studies.
5.2 Separation Methods Coupled with Radioactivity Detection
5.2.1 Thin-Layer Chromatography (TLC) and Radio-TLC
TLC provides a rapid means of checking purity and identity. After developing a TLC plate, the radioactive zones can be visualised by:
- Autoradiography – exposing the plate to X-ray film or phosphor screens.
- Radio-TLC scanners – passing the plate under a detector (scintillation or proportional counter).
Radio-TLC is ideal for quick checks during synthesis but has lower resolution compared to HPLC.
5.2.2 High-Performance Liquid Chromatography (HPLC) with Radiodetection
HPLC is the workhorse for purification and analysis of radiolabelled compounds. A radiometric detector placed after the UV or MS detector measures the radioactivity in each eluted fraction. This allows simultaneous confirmation of chemical identity (via UV or MS) and radiochemical purity.
5.2.3 Gas Chromatography (GC) with Radiodetection
GC can be coupled to a combustion interface, converting the eluted compound into CO₂, which is then trapped in a scintillation cocktail for detection. This is valuable for volatile or thermally stable compounds.
5.3 Detection Methods for ¹⁴C
5.3.1 Liquid Scintillation Counting (LSC)
This is the most common quantitative method for measuring ¹⁴C. The sample is dissolved or suspended in a scintillation cocktail containing fluor molecules. Beta particles from ¹⁴C decay transfer energy to the fluor, producing photons that are detected by photomultiplier tubes.
Advantages:
- High sensitivity (down to a few disintegrations per minute).
- Suitable for solids, liquids, and biological samples.
Limitations: - Requires careful selection of cocktails to minimise quenching effects.
5.3.2 Accelerator Mass Spectrometry (AMS)
AMS is an ultra-sensitive method capable of detecting attomole levels of ¹⁴C. It measures isotope ratios directly (¹⁴C/¹²C), making it independent of radioactive decay. This is particularly useful for human microdosing studies where the administered radioactivity is extremely low.
5.3.3 Autoradiography and Phosphor Imaging
Autoradiography allows visual mapping of radioactivity distribution in thin-layer samples, gels, or tissue sections. Phosphor imaging plates provide higher resolution and sensitivity than traditional film.
5.4 Purity Assessment
Radiochemical purity is expressed as the proportion of total radioactivity associated with the desired compound. Purity levels above 95% are generally required for in vivo studies. Radiochemical impurities can arise from incomplete reactions, degradation, or isotopic exchange.
Chemical purity is assessed using standard analytical tools such as NMR, MS, and UV spectroscopy, often in combination with chromatography.
5.5 Determination of Specific Activity
Specific activity is determined by measuring both the radioactivity (via LSC or AMS) and the chemical concentration (via UV absorbance, MS, or gravimetric methods). This value is critical for interpreting tracer study results, as it allows conversion of measured radioactivity into molar quantities.
5.6 Stability Testing
Radiolabelled compounds can undergo radiolysis, where emitted beta particles cause molecular fragmentation. Stability testing under storage and experimental conditions helps predict shelf-life. Common approaches include:
- Incubation under accelerated ageing conditions (e.g., elevated temperature or light exposure).
- Periodic purity checks via radio-HPLC or radio-TLC.
Key Points: Analytical techniques for carbon-14 radiolabelling combine high-resolution separation with sensitive radioactivity detection. Methods such as radio-HPLC, LSC, and AMS ensure accurate assessment of purity, identity, and specific activity, providing a solid analytical foundation for downstream applications.
The next section will explore applications of carbon-14 radiolabelling, showing how these analytical capabilities are put to use in pharmaceuticals, environmental science, and mechanistic chemistry.
6 – Applications of Carbon-14 Radiolabelling
6.1 Introduction
Carbon-14 radiolabelling is used in a broad spectrum of scientific disciplines. Its unique combination of long half-life, chemical equivalence to stable carbon, and low-energy beta emission enables sensitive detection in complex systems without altering the behaviour of the labelled molecule.
Applications can be broadly grouped into pharmaceutical research, environmental science, and fundamental chemistry and biochemistry. In each area, the objectives, experimental design, and detection methods vary, but the principle remains the same: use the ¹⁴C label to trace the fate and transformations of a molecule over time.
6.2 Drug Metabolism and Pharmacokinetics
In pharmaceutical research, carbon-14 labelling is most often used in ADME studies—Absorption, Distribution, Metabolism, and Excretion.
6.2.1 Mass Balance Studies
A drug candidate is synthesised with a ¹⁴C label, often in a metabolically stable position. After administration to animals or humans, biological samples (urine, faeces, blood, breath) are collected over time. The total recovered radioactivity is compared to the administered dose, providing a mass balance that accounts for all drug-derived material.
6.2.2 Metabolite Identification
Radiolabelled drugs allow the detection of minor metabolites that may be difficult to identify by mass spectrometry alone. Radio-HPLC is used to separate and quantify metabolites, which are then structurally characterised using MS and NMR.
6.2.3 Human Microdosing
In early-phase clinical research, ultra-low doses of ¹⁴C-labelled drugs (typically <100 micrograms) are given to human volunteers to study pharmacokinetics without significant pharmacological effect. The extremely low levels of ¹⁴C are detected by accelerator mass spectrometry (AMS). This approach reduces risk and cost in drug development.
6.3 Environmental Fate Studies
Regulatory agencies require detailed information on the environmental fate of pesticides, industrial chemicals, and other potential pollutants. Carbon-14 labelling is a key tool for these studies.
6.3.1 Soil Degradation
A ¹⁴C-labelled pesticide is applied to soil under controlled conditions. The decline of the parent compound and appearance of metabolites are tracked over time, often under varying temperature, moisture, and light conditions.
6.3.2 Leaching and Run-off Studies
Radiolabelled chemicals can be monitored in leachate from soil columns or in run-off water from field simulations, helping assess potential contamination of groundwater.
6.3.3 Bioaccumulation Studies
In aquatic systems, the uptake of ¹⁴C-labelled compounds by fish, invertebrates, or plants can be quantified, providing data for risk assessments.
6.4 Mechanistic Studies in Chemistry and Biochemistry
6.4.1 Reaction Pathway Elucidation
By incorporating a ¹⁴C label into a specific carbon atom of a reactant, chemists can track its position in the products. This helps reveal mechanisms such as rearrangements, cleavage patterns, or isotopic scrambling.
6.4.2 Kinetic Isotope Effect Measurements
While ¹⁴C kinetic isotope effects are smaller than those for lighter isotopes, careful measurement can still provide mechanistic insights, especially in enzyme-catalysed reactions.
6.4.3 Enzyme Pathway Mapping
In metabolic biochemistry, ¹⁴C-labelled substrates are used to follow the flow of carbon through enzymatic pathways, such as the citric acid cycle or biosynthetic routes in microorganisms.
6.5 Tracing in Ecology and Geoscience
Although most modern ecological studies use stable isotopes, ¹⁴C labelling still finds occasional use in carbon cycle tracing experiments, particularly where high sensitivity is needed. Examples include:
- Measuring primary production in aquatic systems via ¹⁴C-bicarbonate uptake.
- Tracking carbon flow in soil microbial communities.
Key Points: The versatility of carbon-14 radiolabelling lies in its ability to follow molecular fate across disciplines—from determining the metabolic profile of a new drug to monitoring the breakdown of pollutants in soil and water, or dissecting biochemical pathways.
The next section will present case studies, showing specific examples of these applications in action, including pharmaceutical, environmental, and biochemical research scenarios.
7 – Case Studies
7.1 Introduction
Case studies illustrate the practical application of carbon-14 radiolabelling principles, providing examples of how theory translates into real-world research. The following examples cover three main domains: pharmaceuticals, environmental science, and biochemical pathway elucidation.
7.2 Pharmaceutical Case Study – Mass Balance and Metabolite Profiling
Background
A pharmaceutical company developing a novel anti-inflammatory drug needed to complete a human mass balance study to satisfy regulatory requirements. The molecule had a carboxylic acid functional group and an aromatic ring system.
Radiolabelling Strategy
The label was introduced at the carboxyl carbon via a [¹⁴C]CO₂ carboxylation step early in the synthesis. This position was chosen because it was metabolically stable; metabolic cleavage at this point was unlikely, ensuring that the label would remain associated with all drug-related material in the body.
Study Design
Healthy volunteers received a single oral dose of the ¹⁴C-labelled drug. Biological samples (urine, faeces, blood) were collected over 10 days.
Analytical Approach
Total radioactivity in each sample was measured by liquid scintillation counting (LSC). Radio-HPLC with UV and MS detection was used to separate and identify metabolites.
Results
- 92% of the administered radioactivity was recovered: 70% in urine, 22% in faeces.
- The parent drug accounted for 60% of the radioactivity in plasma.
- Two minor metabolites (each <5% of total radioactivity) were identified, both resulting from aromatic hydroxylation.
Conclusion
The study provided a complete mass balance and metabolite identification, meeting regulatory requirements and supporting the drug’s safety profile.
7.3 Environmental Science Case Study – Pesticide Soil Degradation
Background
An agrochemical company needed to evaluate the environmental persistence of a new herbicide before market approval.
Radiolabelling Strategy
The ¹⁴C label was placed on the central heterocyclic carbon, which was stable under environmental conditions, ensuring that any breakdown products could be traced back to the original molecule.
Study Design
Labelled herbicide was applied to three soil types in controlled laboratory conditions. Samples were taken over six months to monitor degradation under aerobic conditions.
Analytical Approach
Soil extracts were analysed by radio-HPLC to quantify the parent compound and metabolites. Volatile breakdown products were trapped and measured separately. Bound residues in the soil were quantified by combustion to ¹⁴CO₂ followed by LSC.
Results
- The herbicide half-life ranged from 45 to 90 days, depending on soil type.
- Two major metabolites were identified, resulting from oxidative cleavage of the heterocycle.
- Approximately 15% of the applied radioactivity became bound to soil organic matter.
Conclusion
The data demonstrated moderate persistence and formation of non-extractable residues, information essential for regulatory risk assessment.
7.4 Biochemical Pathway Case Study – Microbial Carbon Metabolism
Background
A microbiology research group sought to understand how a soil bacterium metabolised a particular aromatic pollutant.
Radiolabelling Strategy
The pollutant was synthesised with a ¹⁴C label in the aromatic ring. This position was chosen because ring cleavage would be a key step in degradation, allowing the label to be detected in downstream metabolites or CO₂.
Study Design
Cultures of the bacterium were grown in minimal medium with the labelled pollutant as the sole carbon source. Samples of culture medium and gas phase were collected over 14 days.
Analytical Approach
Radio-HPLC was used to analyse soluble metabolites. Evolved CO₂ was trapped in a base solution and counted by LSC.
Results
- Within three days, the parent compound disappeared from the medium.
- An intermediate dihydrodiol metabolite accumulated, then decreased as CO₂ levels increased.
- By day 14, 75% of the original radioactivity was recovered as ¹⁴CO₂.
Conclusion
The data confirmed that the bacterium used an initial dioxygenase step to open the aromatic ring, followed by complete mineralisation to CO₂.
Key Points: These case studies demonstrate the adaptability of carbon-14 radiolabelling: from quantifying drug clearance in humans, to assessing chemical breakdown in soils, to mapping microbial metabolic pathways. In each case, success depended on selecting the correct labelling position, applying sensitive analytical methods, and interpreting the radioactivity data within the system’s context.
The next section will address safety, handling, and waste management—critical considerations for any laboratory working with ¹⁴C-labelled materials.
8 – Safety, Handling, and Waste Management
8.1 Introduction
Working with carbon-14 is relatively safe compared to high-energy radionuclides, but it still requires strict adherence to radiological safety procedures. The principal hazard from ¹⁴C is internal contamination via ingestion, inhalation, or absorption through skin wounds. The low penetration of its beta emissions means external exposure is negligible; however, once inside the body, ¹⁴C-labelled organic compounds distribute through normal biochemical pathways, leading to whole-body irradiation until eliminated.
8.2 Laboratory Design and Access Control
8.2.1 Designated Radioisotope Areas
Radiochemistry work with ¹⁴C should be carried out in areas clearly designated for radioactive use, ideally separated from general chemistry spaces. Key features include:
- Impervious, easily cleanable bench surfaces.
- Clearly labelled work zones and equipment.
- Spill trays and absorbent bench liners.
8.2.2 Ventilation
Work with volatile ¹⁴C compounds (e.g., [¹⁴C]methyl iodide, [¹⁴C]CO₂) must be performed in a fume cupboard or glove box with HEPA and activated carbon filtration to capture airborne radioactivity.
8.2.3 Controlled Access
Only authorised, trained personnel should enter radioisotope areas, and entry should be logged.
8.3 Personal Protective Equipment (PPE)
PPE acts as a physical barrier against contamination. Standard PPE for ¹⁴C work includes:
- Laboratory coats (preferably disposable or dedicated to radioactive work).
- Nitrile or latex gloves, changed frequently.
- Safety glasses or goggles.
- Closed footwear.
When working with volatile forms, respiratory protection may also be required, subject to risk assessment.
8.4 Contamination Monitoring
8.4.1 Direct Monitoring
Although ¹⁴C emits low-energy beta particles that are difficult to detect with standard Geiger counters, liquid scintillation-based swipe tests are highly effective.
8.4.2 Indirect Monitoring
Surfaces, equipment, and PPE should be regularly swabbed with moistened filter paper; the swabs are then analysed by liquid scintillation counting to check for contamination.
8.4.3 Personnel Monitoring
Hands and forearms should be monitored before leaving the laboratory. Personal dosimetry badges are not generally used for ¹⁴C, but biological monitoring (urine assays) may be conducted in high-activity work.
8.5 Handling and Storage Practices
8.5.1 Minimising Exposure
- Work with the smallest practical quantities.
- Use pre-weighed aliquots when possible to avoid repeated handling.
- Keep containers closed when not in use.
8.5.2 storage
Store ¹⁴C-labelled materials in clearly labelled, leak-proof containers, segregated by physical form (solids, liquids, volatiles). Keep in cool, dark conditions to minimise radiolysis.
8.6 Waste Classification and Disposal
8.6.1 Waste Types
¹⁴C waste is classified into several categories:
- Solid waste – contaminated PPE, paper, and plasticware.
- Aqueous liquid waste – water-based solutions.
- Organic liquid waste – scintillation cocktails, solvents.
- Gaseous waste – volatile ¹⁴C compounds.
8.6.2 Segregation
Waste streams must be kept separate to allow appropriate disposal routes. Containers should be labelled with isotope, activity, date, and the responsible person.
8.6.3 Disposal Routes
- Solid waste: incineration in licensed facilities.
- Liquid waste: decay storage (if activity is low) or incineration.
- Volatile waste: trapping in carbon filters before disposal.
8.7 Spill Response
In the event of a spill:
- Alert others and restrict access.
- Wear appropriate PPE before clean-up.
- Contain the spill using absorbent material.
- Decontaminate surfaces with detergent or ethanol.
- Monitor the area to ensure contamination removal.
- Dispose of clean-up materials as radioactive waste.
Key Points: The safety profile of carbon-14 is favourable compared to higher-energy radionuclides, but it requires rigorous contamination control, careful handling, and adherence to waste disposal regulations. With appropriate infrastructure, training, and procedures, ¹⁴C radiochemistry can be performed safely and effectively.
The next section will address regulatory and ethical considerations, outlining the frameworks that govern the use of carbon-14 in laboratory, clinical, and environmental contexts.
9 – Regulatory and Ethical Considerations
9.1 Introduction
The use of carbon-14 in research and industry is subject to national and international regulations designed to protect workers, the public, and the environment from radiological hazards. Compliance is not just a legal requirement—it also ensures that research results are scientifically credible and ethically defensible. This section outlines the regulatory frameworks, licensing processes, and ethical principles relevant to ¹⁴C radiolabelling in laboratory, clinical, and environmental contexts.
9.2 International Regulatory Frameworks
9.2.1 International Atomic Energy Agency (IAEA)
The IAEA sets global safety standards for radiation protection and radioactive waste management. While it does not issue operational licences, its guidelines influence national regulations.
9.2.2 International Commission on Radiological Protection (ICRP)
The ICRP provides recommendations on dose limits and exposure control. For occupational exposure, the general annual limit for effective dose is 20 mSv averaged over five years, with no single year exceeding 50 mSv. For ¹⁴C, internal dose coefficients are specified for different chemical forms.
9.2.3 United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR)
UNSCEAR evaluates the global impact of radiation and informs policy decisions, including those affecting radionuclide use.
9.3 National Regulatory Bodies
Specific requirements vary by country:
- United Kingdom – Environment Agency (EA) and Health and Safety Executive (HSE) regulate radioactive substances, with licensing under the Environmental Permitting Regulations and Ionising Radiations Regulations 2017 (IRR17).
- United States – Nuclear Regulatory Commission (NRC) and Agreement State agencies oversee isotope use under Title 10 of the Code of Federal Regulations.
- European Union – Euratom Treaty framework and member-state authorities regulate licensing, waste management, and dose limits.
Licences typically specify isotope limits, authorised locations, waste management protocols, and reporting obligations.
9.4 Licensing and Record Keeping
Before working with ¹⁴C, organisations must obtain a licence covering:
- The isotopes to be used and the maximum quantities.
- Approved storage facilities and laboratories.
- Waste disposal arrangements.
Record keeping is essential and usually includes:
- Inventory logs of isotope acquisition, use, and disposal.
- Experimental records linking isotope use to authorised projects.
- Waste transfer documentation.
These records must be available for inspection by regulatory authorities.
9.5 Ethical Considerations in Human Studies
9.5.1 Informed Consent
Participants in human studies involving ¹⁴C-labelled compounds must receive clear, understandable information about:
- The purpose of the study.
- The amount of radioactivity involved.
- Potential risks and benefits.
9.5.2 Dose Minimisation
Ethical review boards require researchers to use the smallest possible radioactive dose consistent with achieving the study objectives. For microdosing studies, doses are often in the range of a few kilobecquerels, far below levels associated with health risk.
9.5.3 Compliance with Good Clinical Practice (GCP)
All clinical studies must follow GCP guidelines, ensuring ethical treatment, data integrity, and regulatory compliance.
9.6 Ethical Considerations in Environmental Studies
When releasing ¹⁴C-labelled compounds into controlled environmental systems (e.g., soil microcosms, aquatic mesocosms), ethical and regulatory review ensures:
- Containment and prevention of environmental contamination.
- Safe disposal of all study materials.
- Avoidance of unintended ecological harm.
Field studies involving environmental release of ¹⁴C are rare and require exceptional justification and stringent controls.
Key Points: Regulatory compliance in carbon-14 radiolabelling work is non-negotiable, covering licensing, record keeping, dose limits, and waste disposal. Ethical considerations—particularly in human and environmental research—demand transparency, risk minimisation, and adherence to international best practice.
The final section will explore future directions, looking at emerging trends, alternative labelling methods, and the role of advanced analytical and computational tools in radiotracer research.
10 – Future Directions
10.1 Introduction
Carbon-14 radiolabelling has been a cornerstone of tracer research for more than eight decades. While the fundamental principles remain the same, advances in synthesis, detection, and computational modelling are shaping how this technique will be used in the future. This section considers the emerging trends likely to influence the next generation of ¹⁴C research, as well as the challenges and opportunities they present.
10.2 Advances in Synthetic Methodology
10.2.1 Late-Stage Radiolabelling
Recent progress in catalysis has enabled the direct incorporation of ¹⁴C into complex molecules at a late stage of synthesis. These methods reduce synthetic steps, save isotope material, and accelerate preparation of labelled drug candidates. Examples include:
- Transition-metal-catalysed carbonylation using [¹⁴C]CO under mild conditions.
- Photoredox-mediated carboxylations with [¹⁴C]CO₂.
10.2.2 Flow Radiochemistry
Flow chemistry offers enhanced safety, reproducibility, and efficiency. In ¹⁴C labelling, microreactors allow precise control of reagent quantities, minimising waste and operator exposure.
10.2.3 Biocatalytic Labelling
Enzymes are increasingly being explored for introducing ¹⁴C into biomolecules. Enzymatic carboxylases and methyltransferases can achieve regioselective labelling under mild, aqueous conditions.
10.3 Advances in Detection Technology
10.3.1 Accelerator Mass Spectrometry (AMS)
AMS will continue to expand the use of ¹⁴C in ultra-low-dose studies, particularly in pharmacokinetic microdosing. Improved instrumentation offers faster throughput, higher precision, and reduced sample preparation times.
10.3.2 Integrated LC–AMS Systems
Coupling AMS directly to liquid chromatography could streamline metabolite profiling, eliminating the need for fraction collection and offline analysis.
10.3.3 Digital Autoradiography
High-resolution phosphor imaging and digital detectors now allow real-time monitoring of ¹⁴C-labelled compounds in complex systems, from chromatograms to whole-body autoradiograms.
10.4 Alternative and Complementary Labelling Strategies
While ¹⁴C offers unmatched stability for long-term tracing, alternative isotopes and techniques are increasingly being integrated into research programmes:
- Stable isotopes (¹³C, ²H, ¹⁵N) provide non-radioactive labelling for NMR and MS-based studies.
- Positron-emitting isotopes (¹¹C, ¹⁸F) enable real-time imaging by positron emission tomography (PET), although their short half-lives limit experimental duration.
- Dual-labelling approaches combine ¹⁴C with another isotope to simultaneously track different molecular regions or processes.
10.5 Computational and Data-Driven Approaches
The integration of computational chemistry, bioinformatics, and AI is expected to transform radiolabelling research:
- Predictive metabolism modelling can identify optimal labelling sites before synthesis.
- In silico reaction planning helps design efficient labelling routes with minimal isotope loss.
- Automated data interpretation in ADME and environmental studies can accelerate reporting and regulatory submission.
10.6 Sustainability and Regulatory Evolution
As environmental and occupational safety regulations tighten, future radiolabelling research will emphasise:
- Reducing the amount of isotope used through improved detection sensitivity.
- Developing greener synthesis methods with minimal solvent and energy use.
- Enhancing waste minimisation and recycling of labelled intermediates where possible.
10.7 Long-Term Outlook
Carbon-14 radiolabelling will remain indispensable for certain applications—particularly where long-term stability, metabolic fidelity, and ultra-trace detection are required. However, its role will evolve, increasingly complemented by non-radioactive isotopes, high-sensitivity detectors, and digital workflow integration. In the decades ahead, researchers will have a more sophisticated toolbox in which ¹⁴C remains a key, but not exclusive, option.
Key Points: The future of carbon-14 radiolabelling lies in marrying traditional tracer science with modern synthetic chemistry, advanced analytics, and computational intelligence. By embracing innovation while maintaining rigorous safety and ethical standards, ¹⁴C research will continue to deliver unique insights into chemical, biological, and environmental processes.
Conclusion
Carbon-14 radiolabelling remains one of the most versatile and informative tracer techniques available to scientists, combining chemical indistinguishability with stable detection over extended timescales. From drug metabolism and pharmacokinetic studies to environmental fate assessments and mechanistic investigations in chemistry and biochemistry, its applications span multiple disciplines and regulatory contexts. The continued development of synthetic strategies, analytical technologies, and computational tools is expanding the scope, efficiency, and safety of ¹⁴C research. Looking forward, the integration of late-stage labelling, flow radiochemistry, and ultra-sensitive detection methods such as accelerator mass spectrometry will allow researchers to work with smaller quantities of isotope, reducing both cost and radiological risk. By adhering to rigorous safety, ethical, and regulatory standards and by embracing emerging innovations, carbon-14 radiolabelling will continue to provide precise, high-value insights into the movement, transformation, and persistence of molecules in complex systems.
Disclaimer
The information presented in this article is intended solely for educational and informational purposes. Carbon-14 (¹⁴C) radiolabelling involves the use of radioactive materials, which present potential health, safety, and environmental hazards if not handled in accordance with applicable regulations and best laboratory practices. The procedures, techniques, and applications described herein must only be undertaken by qualified personnel working within licensed facilities and following all relevant national and international guidelines. Open MedScience Review, the author(s), and publisher accept no responsibility or liability for any injury, loss, damage, or regulatory non-compliance arising from the use or misuse of the information provided. Readers are responsible for ensuring that any work involving ¹⁴C complies with local licensing, safety, and ethical requirements before commencing such activities.
How to cite: Open MedScience. Carbon-14 Radiolabelling: Theory, Methods, and Applications in Research. Open MedScience Review. 10 August 2025. Available at: https://www.openmedscience.com/carbon-14-radiolabelling-theory-methods-and-applications-in-research
home »