Continuous Glucose Monitoring (CGM) has transformed modern diabetes management, allowing patients to track their glucose trends continuously rather than depend solely on finger‑stick testing. By replacing “snapshot” readings with real‑time data, CGM systems provide deeper insight into glycemic variability and “time in range,” empowering both users and clinicians to make more informed treatment decisions.
According to longitudinal data published in Diabetes, CGM adoption among people with Type 1 diabetes grew from near‑zero use in 2008 to become the primary monitoring method by 2022. This shift has improved patient safety and disease awareness, yet with innovation come new questions about reliability, accuracy, and legal accountability when devices fail.
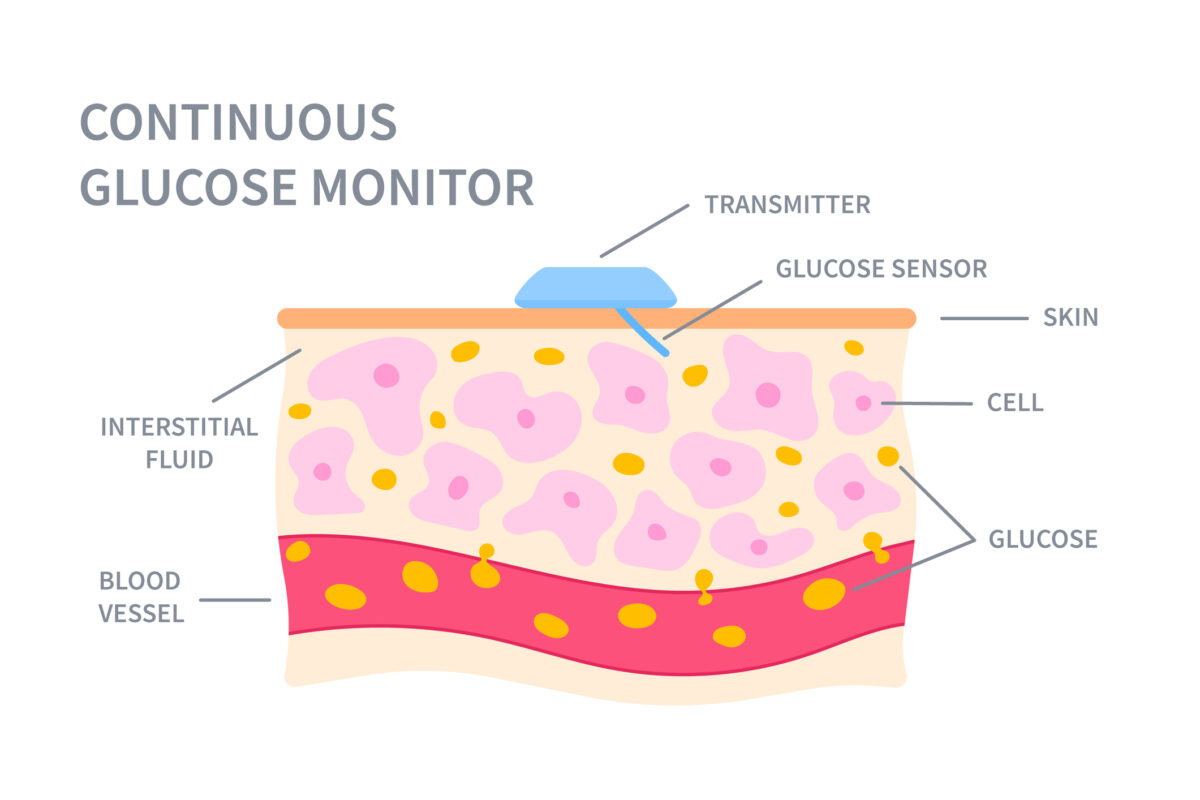
How Continuous Glucose Monitors Work
A CGM measures glucose levels in interstitial fluid (ISF) — the clear liquid surrounding cells. The sensing process involves several steps:
- Sensor Filament: A microscopically thin, enzyme‑coated filament is inserted just under the skin.
- Chemical Reaction: The enzyme, commonly glucose oxidase, reacts with ISF glucose, generating hydrogen peroxide.
- Signal Transduction: This reaction produces a microcurrent proportional to glucose concentration.
- Algorithmic Conversion: A transmitter interprets the current through algorithms into a readable glucose measurement.
Because glucose takes a few minutes to move from blood to interstitial fluid, CGM readings lag behind blood glucose by about five to fifteen minutes. During rapid fluctuations — such as intense exercise or post‑meal spikes — this delay may cause discrepancies. Studies on sensor accuracy show this difference is most evident during sharp glucose changes.
Accuracy Challenges and Regulatory Oversight
Even well-designed CGMs can face accuracy issues:
- Compression lows: Pressure on the sensor can restrict tissue fluid flow, producing false low readings.
- Chemical interference: Medications such as acetaminophen or high-dose vitamin C may affect readings.
- Biofouling: Immune responses can create a protein layer around the sensor, blocking glucose diffusion.
Regulatory authorities like the FDA and the MHRA actively monitor device performance. In cases where failures could cause serious harm, regulators may issue a Class I recall, the highest-level warning.
For example, a false low reading could lead a patient to consume unnecessary carbohydrates or skip insulin doses, potentially causing severe hyperglycemia or diabetic ketoacidosis (DKA).
What Patients Should Know After a Recall
A Class I recall signals a safety issue but does not automatically guarantee compensation. Patients harmed by a recalled CGM must demonstrate causation — that the device malfunction directly caused their injury. Evidence such as preserved sensors, device logs, medical records, and communication with the manufacturer is critical.
How a Medical Product Liability Attorney Can Help
When device issues go beyond technical troubleshooting, consulting a medical product liability attorney is essential. These attorneys specialize in evaluating whether a defective device, manufacturing error, or inadequate warning contributed to a patient’s injury.
A qualified attorney can:
- Investigate regulatory filings and recall notices to determine manufacturer awareness.
- Preserve physical devices and digital data for expert review.
- Collaborate with medical experts to show how false readings affected treatment decisions.
- Pursue compensation for medical expenses, lost wages, and pain and suffering.
Liability claims differ from general product complaints — proving negligence or design flaws is required. Legal guidance ensures cases are documented and regulatory requirements are met.
Staying Safe and Informed
CGMs are among the most impactful tools in diabetes care, helping patients manage their disease effectively. Users should:
- Validate device readings, especially if symptoms conflict with CGM data.
- Monitor regulatory alerts and manufacturer updates.
- Seek professional guidance if a malfunction or recall causes harm.
Timely consultation with a medical product liability attorney provides the bridge between technical investigation and patient protection, ensuring accountability lies with manufacturers and safeguarding patient rights.
Disclaimer
This article is provided for general informational purposes only and does not constitute medical or legal advice. Readers should consult a qualified healthcare professional regarding any medical concerns and seek independent legal advice for matters relating to product liability or device recalls. Open MedScience accepts no responsibility for any actions taken based on the information presented.
home » blog » health matters »