Radiotherapy remains one of the most widely used interventions in oncology. A 2025 review published in CA Cancer Journal for Clinicians (PMC/NIH, 2025) estimated that roughly 50% of all cancer patients receive it at some point during their care. For many, it’s a life-saving component of a curative or palliative treatment plan. But for a meaningful subset of long-term survivors, radiation leaves behind a chronic biological injury that doesn’t resolve on its own.
Late radiation tissue injury (LRTI) – sometimes called late effects of radiation therapy – develops months to decades after treatment ends. It manifests as osteoradionecrosis, radiation hemorrhagic cystitis, soft tissue necrosis, bowel or rectal injury from pelvic radiotherapy, and impaired wound healing in previously irradiated fields. The common thread is progressive vascular damage and hypoxia in tissue that was once adequately perfused. Conventional wound care reaches its limit here because the underlying problem isn’t just surface-level – it’s a failure of oxygen delivery at the tissue level.
Hyperbaric oxygen therapy (HBOT) is one of the few adjunctive interventions with a formal evidence base for this population. The Undersea and Hyperbaric Medical Society (UHMS) lists late radiation tissue injury among its 14 approved indications, placing it in the same category as carbon monoxide poisoning and arterial gas embolism. This article examines the pathophysiological rationale, the clinical evidence across specific LRTI subtypes, and what clinicians and patients should understand about access to appropriate treatment.
What is Hyperbaric Oxygen Therapy?
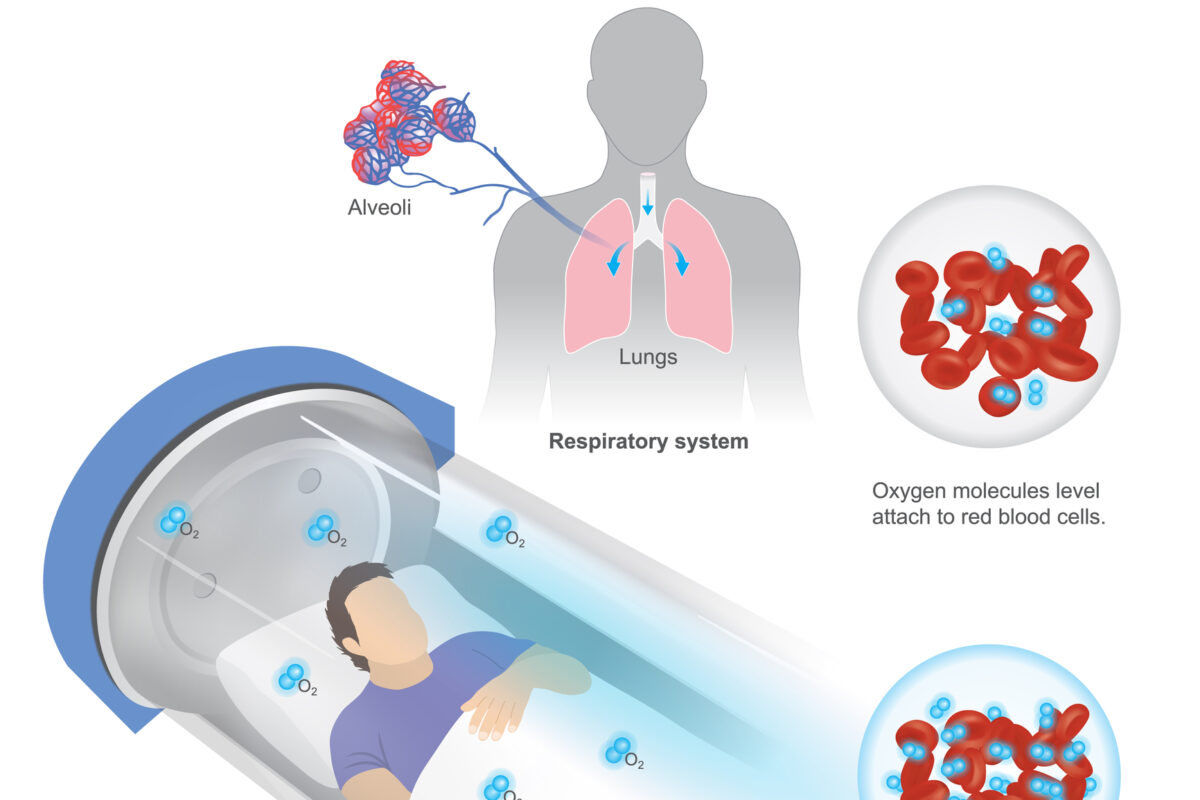
In a hyperbaric oxygen session, the patient breathes 100% oxygen inside a pressurised chamber – typically at 2.0 to 2.4 atmospheres absolute (ATA) – for sessions lasting approximately 90 to 120 minutes. At sea level, dissolved plasma oxygen sits at roughly 3 mL/L. Under HBOT conditions, that figure rises to approximately 60 mL/L, allowing oxygen to reach tissues via plasma transport independent of haemoglobin. For irradiated tissue with damaged vasculature, this distinction is clinically significant.
UHMS currently recognises 14 approved indications for HBOT, including late radiation tissue injury. The NIH Bookshelf chapter on delayed radiation injury (NCBI, 2024) notes that approximately one-third of all HBOT patients in the United States are receiving treatment specifically for late effects of radiation therapy – making LRTI one of the most common indications in clinical hyperbaric practice.
Patients researching equipment access will find that options for a hyperbaric chamber for sale span a wide range of pressurisation levels and intended use categories. Clinical-grade systems certified for UHMS-approved indications require at least 2.0 ATA capability. Lower-pressure wellness units – typically operating below 1.5 ATA – fall outside the evidence base for medical conditions like LRTI. This distinction matters: the therapeutic effects described throughout this article are derived from protocols using clinically validated pressures, not from wellness-grade devices.
The approved indications framework is detailed in the StatPearls hyperbaric therapy for wound healing chapter (NCBI/NIH, 2024), which also outlines the clinical criteria used by treating physicians to determine eligibility for insurance-covered sessions.
How HBOT Addresses the Pathophysiology of Radiation Injury
Radiation injury doesn’t simply damage cells at the time of treatment. It initiates a process of progressive endarteritis obliterans – the obliteration of small blood vessels in irradiated fields through fibrous thickening and vessel wall damage. Over time, this creates a hypoxic, hypovascular, hypocellular tissue environment: the classic triad described in the radiation wound literature. Conventional wound care struggles here because fibroblasts, which are responsible for matrix remodelling and wound closure, don’t function well in low-oxygen conditions.
HBOT interrupts this cycle through several mechanisms. First, the dramatic rise in dissolved plasma oxygen restores tissue oxygen tension in areas where damaged vasculature can no longer deliver an adequate supply. Second, sustained hyperoxia stimulates angiogenesis – the growth of new capillaries into hypoxic tissue – through modulation of vascular endothelial growth factor (VEGF) and hypoxia-inducible factor 1-alpha (HIF-1α) signalling. New vessel formation gradually rebuilds perfusion capacity in the irradiated field.
At the molecular level, the combined hyperbaric-hyperoxic environment also alters gene expression. A 2024 review published in PMC (NIH, 2024) found that HBOT can modify the expression of approximately 40% of protein-coding genes, producing anti-inflammatory, anti-apoptotic, and regenerative effects at the cellular level. This epigenetic dimension helps explain why HBOT produces durable tissue changes rather than just transient oxygen supplementation.
HBOT also reduces pro-inflammatory cytokines – including TNF-alpha and IL-6 – and attenuates oxidative stress, both of which drive chronic radiation damage. Understanding late effects from external beam radiotherapy helps clarify why this multi-mechanism approach is necessary: radiation injury is not a single-pathway problem, and it doesn’t respond to single-pathway solutions.
Clinical Evidence for HBOT in Radiation-Related Conditions
The evidence for HBOT in LRTI is strongest for three well-studied subtypes: osteoradionecrosis, radiation-induced hemorrhagic cystitis, and soft-tissue wound complications.
Osteoradionecrosis
Osteoradionecrosis of the jaw – a frequent and debilitating complication in head and neck cancer survivors – is among the better-studied HBOT indications. Radiation destroys the vascularity of bone tissue; the mandible is particularly vulnerable given the radiation fields used for oral and pharyngeal cancers. HBOT is used both prophylactically (before dental procedures in irradiated fields) and therapeutically (once necrosis has developed). A 2024 systematic review in PubMed assessed HBOT as an adjunct for head and neck cancer survivors with radiation complications and found evidence supporting its use in preventing and treating bony necrosis in this population.
Radiation Hemorrhagic Cystitis
Pelvic radiotherapy for bladder, prostate, cervical, and colorectal cancers can produce radiation hemorrhagic cystitis – a painful, often refractory complication characterised by mucosal damage, bleeding, and recurrent urinary symptoms. A 2024 systematic review and meta-analysis (PMC/NIH, 2024) examined HBOT for radiation-induced cystitis across multiple cohorts and found response rates ranging from 67% to 100%, with HBOT demonstrating consistent clinical benefit where conventional management had failed.
Soft Tissue Necrosis and Wound Complications
Radiation impairs wound healing by destroying the vascular architecture that repair processes depend on. This becomes clinically significant after surgical procedures in previously irradiated areas – particularly reconstructive surgery following mastectomy or head and neck cancer resection. A 2024 systematic review published in PubMed found that adjunctive HBOT is both safe and effective for hard-to-heal wounds, with specific benefit noted for ischemic and irradiated tissues.
Across all three sub-types, the consistent pattern is that HBOT doesn’t replace standard wound care or surgical management – it restores the biological conditions that make those interventions work. Clinicians working in precision radiation therapy planning will recognise this as an extension of the same logic: treatment efficacy depends on tissue biology, not just technique.
Patient Considerations and Access
The standard HBOT protocol for radiation injury involves approximately 30 to 40 sessions, each lasting 90 to 120 minutes, at 2.0 to 2.4 ATA. Most clinical centres deliver this over six to eight weeks. Sessions take place in monoplace (single-patient) or multiplace (multiple-patient) chambers; for LRTI treatment, clinical-grade monoplace systems that meet UHMS standards are the relevant technology.
Adverse effects are generally mild. Barotrauma – discomfort from pressure equalisation in the ears or sinuses – is the most common complaint and is typically manageable. Transient myopia can occur with extended treatment courses but resolves after treatment ends. Oxygen toxicity seizures are rare at standard therapeutic pressures. The primary contraindication is an untreated pneumothorax. Overall, the safety profile of HBOT under approved protocols is well established.
On insurance coverage: in the United States, Medicare and many private insurers cover HBOT for UHMS-approved indications, including radiation injury, though documentation requirements and prior authorisation processes vary by payer. Patients should expect their treating hyperbaric physician to provide clinical justification tied to the specific approved indication.
The broader market context is worth noting for those tracking healthcare trends. Maximise Market Research (2025) valued the global HBOT market at USD 4.41 billion in 2024, projecting it to reach USD 8.16 billion by 2032 at a compound annual growth rate of 8%. This growth reflects rising patient interest and expanding outpatient infrastructure – but it also reflects the proliferation of non-clinical, low-pressure wellness devices that don’t meet the threshold for treating conditions like LRTI. Clinicians advising patients should be clear: a 1.3 ATA soft-shell chamber is not a medical HBOT unit, and the evidence reviewed here doesn’t apply to it.
For patients managing post-treatment complications across radiation modalities, understanding targeted radionuclide therapy provides a broader context for how radiation dose delivery shapes late tissue responses – and why some patients are more vulnerable to LRTI than others.
Conclusion
HBOT is a formally approved, mechanistically well-characterised adjunctive therapy for late radiation tissue injury. Its ability to restore dissolved oxygen delivery to hypoxic, fibrotic, post-irradiation tissue addresses the core pathophysiology of LRTI – the hypovascular, hypoxic, hypocellular triad – in a way that topical wound care and systemic medications cannot.
The evidence base is strongest for osteoradionecrosis, radiation-induced hemorrhagic cystitis, and soft-tissue wound complications, where multicenter data consistently show meaningful clinical benefit. Gaps remain: larger randomised controlled trials with standardised protocols would strengthen the evidence for some sub-indications, and patient selection criteria are still being refined through ongoing clinical trial activity. But the existing body of evidence – including multiple 2024 systematic reviews and meta-analyses – is sufficient to support HBOT as a legitimate consideration in LRTI management, not a speculative one.
For radiation oncologists, survivorship clinicians, and specialists managing post-treatment complications, the key clinical question isn’t whether HBOT works for radiation injury. The evidence for specific indications like cystitis and osteoradionecrosis is clear. The practical questions are patient selection, timing of referral, and ensuring access to clinical-grade facilities with appropriately pressurised systems. Early referral – before irreversible fibrosis is established – generally produces better outcomes. That’s a practical message worth carrying into multidisciplinary survivorship care planning.
Disclaimer: This article is provided for general informational and educational purposes only and does not constitute medical advice, diagnosis, treatment, or a recommendation for any specific therapy, device, clinic, or healthcare provider. Hyperbaric oxygen therapy (HBOT) should only be considered following assessment by a suitably qualified medical professional with experience in the management of radiation-induced tissue injury. Treatment suitability, risks, outcomes, availability, and insurance coverage can vary according to the patient’s medical history, clinical condition, and local healthcare arrangements. References to clinical studies, approved indications, commercial equipment, or market trends are included for context and should not be interpreted as endorsement. Patients should always seek personalised advice from their oncologist, hyperbaric physician, or other relevant healthcare professional before making decisions about care.
home » blog » radiation therapy »