Theragnomics represents a pioneering approach in personalised medicine that combines the targeted diagnostic and therapeutic capabilities of theranostics with the data-driven insights of radiomics. This innovative concept promises to revolutionise cancer treatment by enabling more precise diagnosis, treatment planning, and monitoring. By leveraging the detailed imaging data that radiomics provides and the customised treatment solutions of theranostics, theragnomics aims to optimise patient care, reduce side effects, and improve overall outcomes.
Introduction to Theragnomics
The field of oncology is on the brink of a transformative era with the advent of theragnomics. This groundbreaking approach synergises the precision of theranostics and the data-rich insights of radiomics. This innovative concept heralds a new age in cancer treatment. It embodies the ultimate goal of personalised medicine: to provide targeted, effective care based on the individual characteristics of each patient’s disease.
Theranostics, a combination of ‘therapy’ and ‘diagnostics’, represents a dual approach to healthcare where diagnostic testing is used to identify specific biological markers (biomarkers) in the body that help select the most effective therapeutic intervention for a particular condition. This method has gained significant traction in the treatment of various cancers, offering a beacon of hope by enabling the use of targeted radiopharmaceuticals. These agents are designed to both visualise and treat malignancies, allowing for a more precise attack on cancer cells while sparing healthy tissues. This specificity enhances the efficacy of treatment and significantly reduces the side effects commonly associated with conventional cancer therapies.
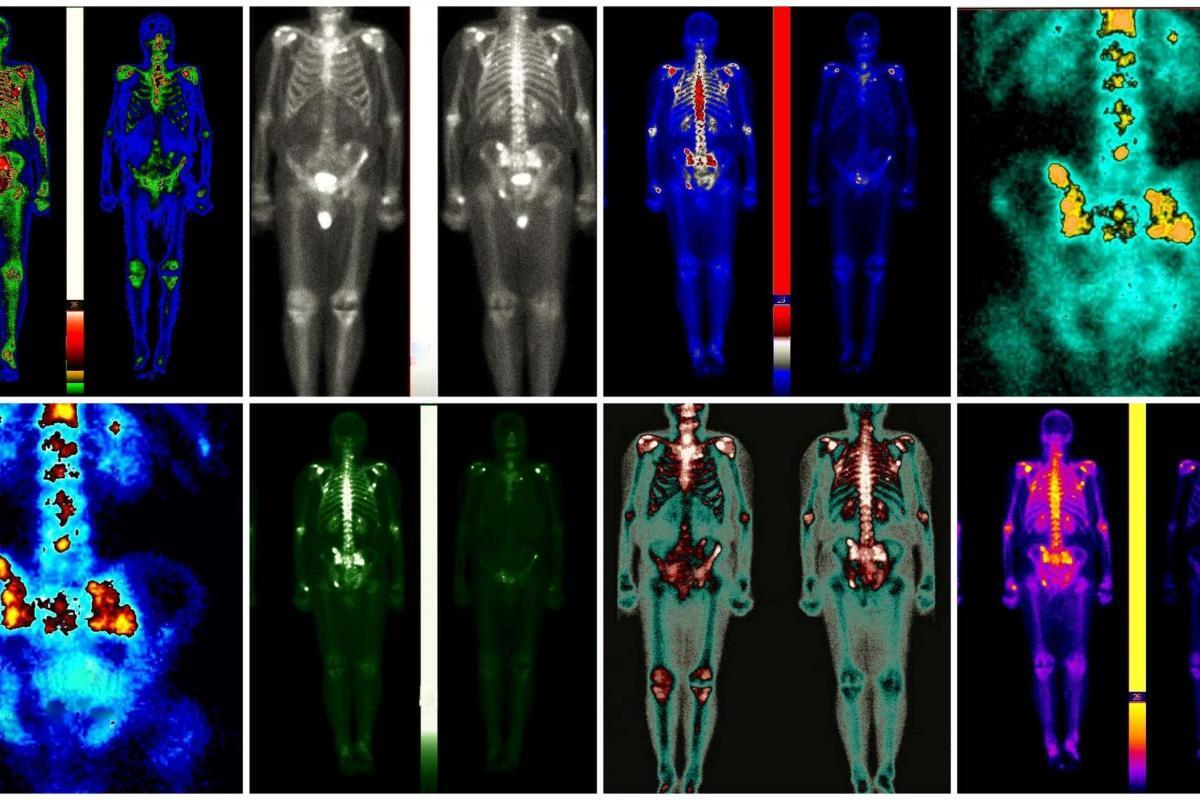
Radiomics, conversely, stands as a testament to the power of modern imaging technology coupled with advanced computational analytics. It involves extracting a vast array of quantitative features from medical images that are not visible to the human eye, such as CT (Computed Tomography), MRI (Magnetic Resonance Imaging), and PET (Positron Emission Tomography) scans. These features can provide detailed information about a tumour’s genetic makeup, behaviour, and prognosis. By analysing this data, radiomics opens new avenues for understanding the complex nature of cancer and tailoring treatment strategies to the individual characteristics of each tumour. This method has shown promise in predicting treatment outcomes, assessing the risk of recurrence, and even uncovering new targets for therapy.
The fusion of theranostics and radiomics into theragnomics represents an evolution in cancer care. By leveraging the diagnostic precision of theranostics to select and administer targeted treatments and employing the analytical depth of radiomics to understand the tumour’s phenotype and predict its behaviour, theragnomics offers a holistic, data-driven approach to oncology. This strategy aims to improve treatments’ accuracy and customise care to the patient’s specific disease profile, thereby maximising efficacy and minimising unnecessary exposure to potentially harmful treatments.
Theragnomics stands at the forefront of personalised medicine, promising to transform the landscape of cancer treatment. Its implementation could lead to more accurate prognostic assessments, more effective treatment selections, and the ability to monitor the disease’s response to treatment in real time. This approach enables oncologists to adapt treatment plans as the disease evolves, ensuring that patients always receive the most appropriate therapy for their current condition.
However, the journey towards fully integrating theragnomics into clinical practice is not without its challenges. The complexity of the data involved requires sophisticated analytical tools and a deep understanding of both the biological aspects of cancer and the technical nuances of imaging and data analysis. Furthermore, the development of new theranostic agents and radiomic algorithms must be accompanied by rigorous clinical trials to establish their efficacy and safety.
The potential of theragnomics to revolutionise cancer care is immense. As research in this field advances and technologies continue to improve, theragnomics could become the cornerstone of oncology, offering hope to millions of patients worldwide. Through the precise targeting of cancer cells and the tailored adaptation of treatment strategies, theragnomics paves the way for more effective cancer treatments and embodies the spirit of personalised medicine, where treatment is as unique as the patients themselves.
Theranostics: A Pillar of Personalised Medicine
Theranostics stands as a revolutionary pillar in personalised medicine, embodying an approach that integrates diagnosis with therapy to tailor treatment to the individual’s unique disease profile. This innovative concept leverages specific biological markers, or biomarkers, to accurately diagnose conditions and determine each patient’s most effective treatment strategy. Particularly transformative in the field of oncology, theranostics uses radiolabelled compounds to target and annihilate cancer cells with unprecedented precision, thereby significantly enhancing treatment efficacy and reducing the risk of adverse effects associated with traditional therapies.
At its core, theranostics is about moving beyond the one-size-fits-all model of healthcare. It acknowledges the intricate variability in how individuals respond to treatments based on their genetic makeup, disease characteristics, and other personal factors. By identifying specific biomarkers in a patient’s body, theranostics allows for the selection of a therapeutic agent that is most likely to be effective for their particular condition. This level of specificity is crucial in cancer treatment, where the difference between targeting the right and wrong molecular pathways can mean the difference between life and death.
The role of radiolabelled compounds in theranostics cannot be overstated. These specially designed molecules are tagged with radioactive isotopes that enable both the visualisation of tumour cells through advanced imaging techniques and the delivery of therapeutic radiation directly to the cancer cells. This dual functionality facilitates the early detection of malignancies and allows for the targeted destruction of tumour tissue, minimising damage to healthy cells surrounding the cancer. Such targeted therapy is a significant departure from conventional cancer treatments like chemotherapy and radiotherapy, which often affect both cancerous and healthy tissues, leading to a range of side effects that can severely impact the patient’s quality of life.
The application of theranostics in oncology has already shown promising results in treating various types of cancer, including neuroendocrine tumours, prostate cancer, and thyroid cancer, among others. For instance, in prostate cancer, the use of PSMA (Prostate-Specific Membrane Antigen) ligands labelled with radioactive isotopes has dramatically improved the accuracy of tumour detection and the effectiveness of subsequent treatments. This approach has not only led to better outcomes for patients but has also opened up new avenues for the development of more specific and potent theranostic agents.
Moreover, the advent of theranostics is facilitating a more collaborative and interdisciplinary approach to cancer care, involving nuclear medicine specialists, radiologists, oncologists, and molecular biologists, among others. This cooperative effort is crucial for the continuous improvement and refinement of theranostic strategies, ensuring that they remain at the cutting edge of personalised medicine.
However, the implementation of theranostics faces challenges, including the need for robust clinical trials to validate the efficacy and safety of new theranostic agents, the development of more sophisticated imaging technologies to detect biomarkers, and the need for a regulatory framework that can keep pace with the rapid advancements in this field. Moreover, the cost of developing and administering theranostic treatments can be high, necessitating efforts to make these therapies more accessible to a broader range of patients.
Theranostics represents a cornerstone of personalised medicine, offering a beacon of hope for patients with cancer and other diseases. By marrying diagnostics with therapeutics, theranostics paves the way for treatments that are more effective and kinder, heralding a new era in healthcare where treatments are as unique as the patients themselves. As research progresses and technological advancements continue, theranostics is set to redefine the boundaries of medical treatment, making personalised medicine a reality for more people worldwide.
Radiomics: The Power of Quantitative Imaging
Radiomics represents a revolutionary leap in the field of medical imaging, marking a transition from qualitative interpretations to a quantitative, data-driven understanding of disease characteristics. This cutting-edge approach has significantly enhanced the potential of imaging technologies like CT, MRI, and PET scans, transforming them into comprehensive sources of biometric data. By meticulously extracting and analysing a vast array of features from medical images, radiomics unlocks insights into the tumour’s genetic makeup, aggressiveness, and likely response to various treatments, thereby playing a pivotal role in the advancement of personalised medicine.
The cornerstone of radiomics lies in its ability to probe deeper into the pixel or voxel level of medical images, identifying patterns and features that are imperceptible to the human eye. These features encompass a wide range of data, including the tumour’s shape, texture, and intensity and the spatial relationships between different image regions. Through advanced computational algorithms, radiomics quantifies these characteristics, generating a detailed fingerprint of the tumour that reflects its underlying biology and behaviour.
This wealth of quantitative data provides a more nuanced understanding of the tumour, far beyond what traditional imaging techniques could offer. For instance, the textural analysis can reveal the heterogeneity within a tumour, which has been linked to its aggressiveness and potential resistance to specific treatments. Similarly, the shape and edge features analysis can offer clues about the tumour’s growth patterns and interactions with surrounding tissues. By correlating these radiomic features with genetic and clinical data, researchers and clinicians can gain insights into the molecular pathways driving the disease, opening up new avenues for targeted therapies.
One of the most promising aspects of radiomics is its potential to personalise treatment plans. By predicting a tumour’s response to various treatments based on its radiomic profile, clinicians can tailor therapies to the individual patient. Therefore, choosing the most likely effective options and sparing them from the side effects of less suitable treatments. This approach improves the efficacy of cancer care and enhances patients’ quality of life by avoiding unnecessary or harmful interventions.
Moreover, radiomics has shown considerable promise in monitoring disease progression and predicting patient outcomes. By comparing changes in the radiomic features of a tumour over time, clinicians can assess how well a patient is responding to treatment and make timely adjustments to their therapeutic strategy. This dynamic approach to cancer care ensures that treatments remain aligned with the evolving nature of the disease, potentially improving survival rates and patient well-being.
The predictive power of radiomics also extends to prognostication. Studies have demonstrated that certain radiomic features are strong predictors of patient outcomes, including survival rates and the likelihood of recurrence. These insights can guide decision-making at every stage of the disease, from diagnosis through to post-treatment monitoring, enabling a more proactive and personalised approach to cancer management.
The integration of radiomics into clinical practice faces challenges, including the need for standardisation in image acquisition and analysis protocols, as well as the development of robust predictive models that can be reliably applied across diverse patient populations. However, as research in this field progresses and technological capabilities continue to advance, radiomics is poised to play an increasingly central role in the personalisation of cancer treatment, offering a new horizon in the quest for more effective, patient-centred care.
The Convergence of Theranostics and Radiomics: A New Era of Cancer Care
The integration of theranostics and radiomics, culminating in the innovative field of theragnomics, heralds a transformative era in cancer care. This synergistic convergence represents a quantum leap in oncology, offering a highly personalised approach to treatment that is predicated on an in-depth understanding of the unique characteristics of each tumour. The fusion of these two cutting-edge technologies enables clinicians to devise treatment plans that are not only tailored to the individual’s genetic and molecular profile but are also dynamically adaptable based on real-time data and outcomes.
Theranostics, with its dual capacity for precise diagnosis and targeted therapy, offers a level of specificity in cancer treatment that was previously unattainable. By utilising biomarkers to identify the most suitable therapeutic agents, this approach ensures that each patient receives a treatment specifically designed to target the peculiarities of their tumour. Radiomics complements this by providing a detailed tumour phenotype analysis through advanced imaging techniques. The extraction of vast amounts of data from medical images allows for a comprehensive characterisation of the tumour, including insights into its aggressiveness, genetic makeup, and potential treatment responses.
When these two disciplines merge into theragnomics, the result is a powerful, data-driven approach that enhances the precision of cancer treatments. This integration allows for identifying the most appropriate theranostic agents based on the radiomic profile of the tumour, ensuring that therapies are highly targeted and optimally effective. Moreover, the detailed imaging data provided by radiomics enables ongoing monitoring of the tumour’s response to these treatments, allowing clinicians to make informed decisions about adjusting or changing treatment strategies as necessary.
This dynamic approach to cancer care is revolutionary, offering several key benefits. Firstly, it increases the efficacy of treatments by ensuring that they are perfectly matched to the specific characteristics of the tumour. This not only maximises the potential for successful outcomes but also significantly reduces the likelihood of the tumour developing resistance to treatment. Secondly, by minimising exposure to ineffective or unnecessarily aggressive treatments, theragnomics minimises the risk of adverse side effects, thereby improving the patient’s quality of life during and after treatment.
Furthermore, the real-time adaptability of theragnomics represents a significant advancement in oncology. The ability to monitor treatment effectiveness in real-time and adjust strategies accordingly means that treatments can be continuously optimised to meet the evolving needs of the patient. This not only enhances the chances of treatment success but also offers the potential to extend survival rates and improve overall outcomes for cancer patients.
The potential of theragnomics to revolutionise cancer care is immense, but its implementation is not without challenges. The complexity of integrating and interpreting vast amounts of genetic, molecular, and imaging data requires sophisticated computational tools and a multidisciplinary approach to treatment planning. Moreover, developing new theranostic agents and radiomic algorithms and the need for extensive clinical trials to validate their effectiveness presents significant hurdles.
However, these challenges, the convergence of theranostics and radiomics into theragnomics is a testament to the relentless pursuit of innovation in the fight against cancer. As research in this field progresses and technology advances, theragnomics stands poised to redefine the landscape of oncology, offering hope for more effective, personalised, and adaptive cancer care. This new era of cancer treatment not only promises improved outcomes for patients but also embodies the ultimate goal of personalised medicine: to treat each patient as an individual, with care that is as unique as their DNA.
Personalised Treatment Plans
Theragnomics ushers in a new epoch in oncology, centring on the development of personalised treatment plans that transcend the one-size-fits-all approach historically prevalent in cancer care. This innovative methodology meticulously tailors therapy to align with the intricate biological landscape of each patient’s tumour, leveraging the detailed insights provided by radiomics and the targeted capabilities of theranostics. By scrutinising the genetic and phenotypic nuances of the tumour through advanced imaging and molecular diagnostics, theragnomics enables the identification of the most efficacious therapeutic agents and strategies for each individual case.
This bespoke approach to treatment planning is not merely about enhancing the efficacy of cancer therapy; it also significantly mitigates the risks associated with traditional treatment methods. By focusing on the therapies that are most likely to be effective for the specific tumour type and its characteristics, theragnomics minimises the patient’s exposure to treatments that may not only be ineffectual but could also entail severe side effects. Consequently, this personalised strategy aims to improve clinical outcomes and prioritises the patient’s quality of life, ensuring a treatment pathway that is as minimally invasive and disruptive as possible.
The paradigm shift towards personalised treatment plans through theragnomics represents a milestone in the pursuit of precision medicine in oncology. It embodies a commitment to understanding the unique complexities of each patient’s cancer, promising a future where treatments are not just targeted but genuinely tailored to the individual, maximising the potential for successful outcomes while safeguarding patient well-being.
Improved Patient Outcomes
The advent of theragnomics heralds a promising shift towards enhanced patient outcomes in the area of cancer care. This innovative approach, by ensuring that treatments are precisely targeted and meticulously monitored, holds the potential to impact patient recovery and well-being profoundly. One of the most significant benefits of theragnomics is the potential for increased survival rates. By tailoring treatments to each tumour’s specific genetic and molecular characteristics, theragnomics maximises the effectiveness of therapeutic interventions, thereby improving the chances of successful outcomes and prolonged survival.
Furthermore, theragnomics prioritises the quality of life of patients undergoing cancer treatment. Traditional cancer therapies, while effective to varying degrees, are often associated with a plethora of adverse side effects that can severely impact a patient’s daily life. Theragnomics, through its precision-targeted approach, aims to minimise these unwanted effects. Selecting therapies that are optimally suited to each individual’s cancer profile reduces exposure to treatments that are less likely to be beneficial, or that may cause unnecessary harm. Consequently, patients experience fewer side effects, leading to an overall improvement in their quality of life during and after treatment.
In essence, theragnomics embodies a holistic approach to cancer care, one that not only strives to extend life but also to enhance the living experience of those battling cancer. Its emphasis on accuracy, personalisation, and adaptability paves the way for a future where cancer treatments are more effective and compassionate, taking into account each patient’s physical and emotional well-being.
Future Directions in Theragnomics Research
As the burgeoning field of theragnomics continues to evolve, the trajectory of its research holds the promise of further transforming cancer care. At the intersection of theranostics and radiomics, this promising domain is set for a future where its capabilities could be vastly expanded through dedicated research and technological advancements.
A pivotal area of focus will be the refinement of radiomics. The aim is to develop more sophisticated algorithms capable of extracting even finer details from medical images, thereby enhancing the predictive power of radiomic signatures. This refinement will enable a deeper understanding of tumour heterogeneity and its implications for treatment response, potentially uncovering new biomarkers for disease characterisation.
Parallelly, the development of novel theranostic agents is critical. Future research is expected to broaden the spectrum of these agents, making them capable of targeting a more diverse array of tumour types and molecular pathways. Exploring innovative delivery mechanisms and engineering more potent radiopharmaceuticals could significantly improve the specificity and efficacy of cancer treatments.
Another transformative aspect of future theragnomics research lies in the integration of artificial intelligence (AI) and machine learning (ML). These technologies promise to revolutionise the analysis of complex datasets, from genomic information to radiomic features. AI and ML algorithms could automate the interpretation of data, enhancing the speed and accuracy with which personalised treatment plans are devised. Moreover, these technologies have the potential to identify patterns and correlations beyond human recognition, offering novel insights into cancer biology and treatment response.
The convergence of AI and ML with theragnomics could also facilitate the development of predictive models for treatment outcomes, enabling more informed decision-making. By continuously learning from a vast array of clinical data, these models could predict the efficacy of specific theranostic agents for individual patients, further personalising cancer care.
In conclusion, the future of theragnomics research is bright, with myriad opportunities for innovation and advancement. By honing the precision of radiomics, expanding the arsenal of theranostic agents, and harnessing the power of AI and ML, theragnomics stands on the cusp of redefining oncology. This progressive integration promises to enhance the efficacy and efficiency of cancer treatments and elevate the standard of care to unprecedented levels. As we venture into this new era, theragnomics emerges as a scientific advancement and a beacon of hope, symbolising a future where cancer treatment is as individualised as the patients it aims to cure.
Disclaimer
The content provided in this article, Combining Theranostics and Radiomics: The Emergence of Theragnomics, is intended for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Readers should not rely solely on the information presented here when making healthcare decisions and are encouraged to consult qualified healthcare professionals for guidance tailored to individual circumstances.
Open Medscience does not endorse or guarantee the effectiveness, safety, or suitability of any medical treatments, technologies, or procedures mentioned in this article. While every effort has been made to ensure the accuracy of the content at the time of publication (07 March 2024), ongoing research and developments in the field may result in changes to scientific understanding or clinical practice that are not reflected in this text.
The views expressed are those of the authors and contributors and do not necessarily represent the opinions of Open Medscience or its editorial team. Open Medscience accepts no responsibility for any loss, damage, or injury arising from the use of the information provided.
home » blog » radiotheranostics »