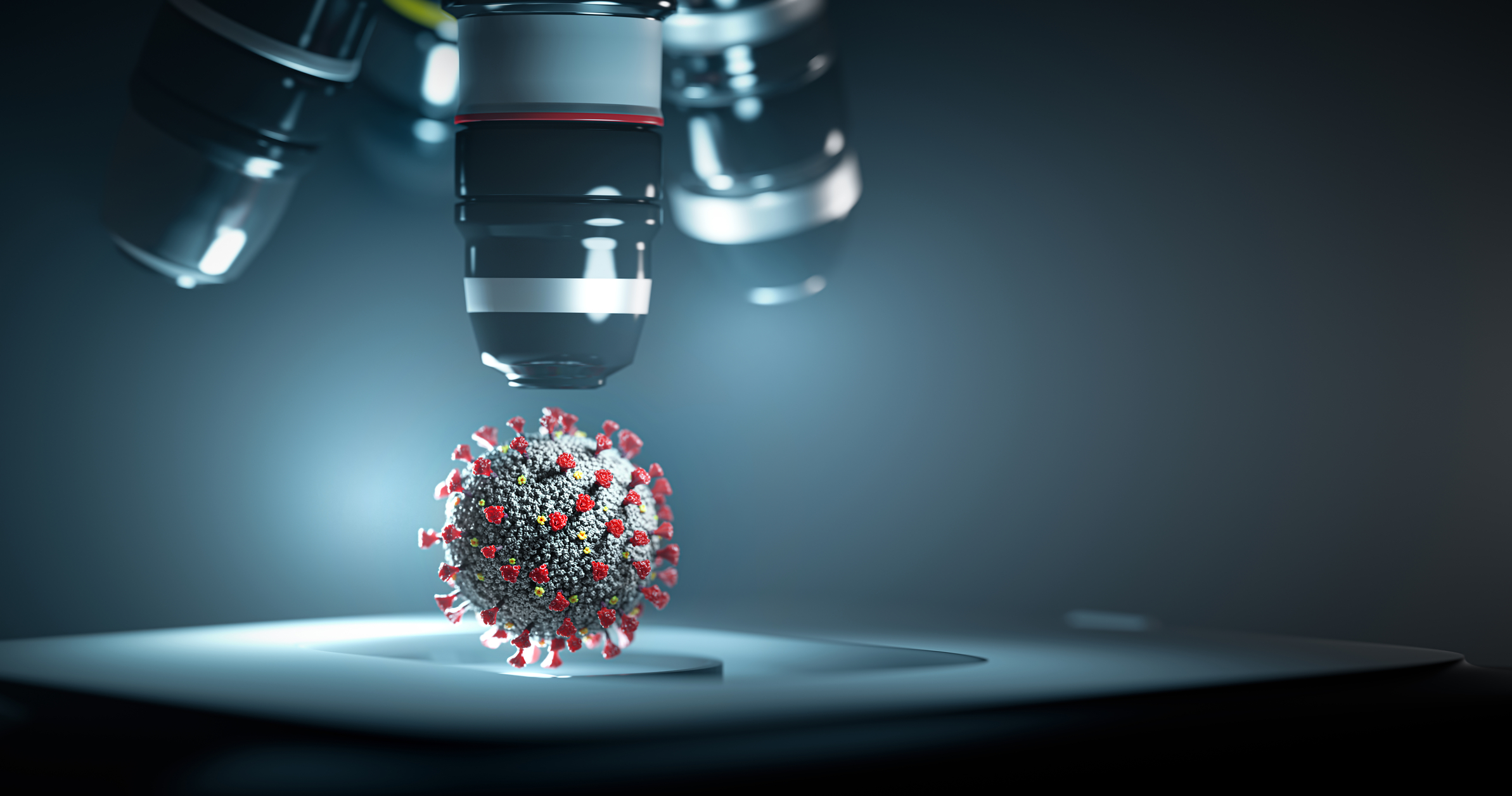
A virus’s structures can be elucidated using the high-resolving power of scanning electron microscopy.
Zooming in on the Enemy: Visualizing SARS-CoV-2 through Electron Microscopy
Virus particles are invisible to the human eye and live in the nanoscale world of biology. However, a virus’s inside structures can be elucidated using the high-resolving power of scanning electron microscopy (SEM).
The visualisation of a virus was only possible through the invention of the first electron microscope in 1931 by Ernst Ruska and Max Knoll. However, in 1939, scientists Ruska, Kausche and Pfankuch became the first to visualise viruses such as the tobacco mosaic virus using the technique of electron microscopy. These developments led to Ruska sharing the 1986 Nobel Prize with Binnig and Rohr for inventing the scanning tunnelling electron microscope.
Electron microscopy has been used to visualise organisms smaller than bacteria and viruses. In the clinical setting, it is valuable in surveilling emerging diseases and potential bioterrorism viruses. Other imaging modalities used to elucidate the structural components of viruses and provide information for treatment and vaccine strategies include immunoelectron microscopy, cryo-electron microscopy and electron tomography.
Other milestones of electron microscopy have enabled the discovery of new viruses. For example, in 1948, electron microscopy could differentiate between the virus that causes smallpox and chickenpox. Another milestone using electron microscopy was its ability in 1952 to obtain the first image of the poliovirus.
The advantage of using electron microscopy to obtain a viral diagnosis is that it does not require biological reagents of specific probes to identify the pathogenic agent. Furthermore, an unknown disease requires a particular reagent to identify the pathogen. Conversely, electron microscopy allows an open view of what is causing the disease compared to molecular tests, which would require an understanding of the agents to determine the correct diagnostic investigation.
EM is a mainstay in detecting new and unusual outbreaks. For example, norovirus (Norwalk agent) was discovered by EM, and EM continues to serve to confirm infection in quality control of molecular techniques.
Electron microscopy was used to identify the viral agent that caused Zaire’s first Ebola virus outbreak in 1976. Also, in 1999, the skin infection trichodysplasia spinulosa was identified by electron microscopy as a polyomavirus in an immunosuppressed patient. Furthermore, the Henipavirus outbreaks in Australia and Asia were first described using electron microscopy in 2003. They identified the lymphocytic choriomeningitis virus that caused fatalities of recipients of organs transplanted from a single donor.
However, electron microscopy was used to identify the severe acute respiratory syndrome (SARS) agent before it was classified as a coronavirus. Also, the cause of the monkeypox outbreaks in the United States in 2003 was discovered by electron microscopy and identified as the poxvirus.
Other methods to identify a virus require growing culture and may be unsuitable for molecular testing because the virus solution has been stored over time. In these cases, electron microscopy does not require a live virus and has been used to identify the variola virus in infected tissue preserved for several decades.
Electron Microscopy and the Battle against Coronavirus: The Key to Understanding its Structure
During the coronavirus outbreak, electron microscopy has been an essential tool for virus detection available to virologists. In the media, we have seen artists’ impressions of the SARS-CoV2 coronavirus. However, electron microscopy has been used to obtain real images of the COVID-19 and shows the SARS-CoV2 virus resembling a small pepperoni pizza.
Viruses are too small to see using conventional microscopes, mainly used to identify bacterial and fungal infections. This is because viruses come in different sizes; the most significant ones are about 500 nm. So in the conventional microscope, they would appear as dots. However, the smallest is around 20 nm, so you could never see them in a light microscope.
The SARS-CoV2 is detected using a PCR test that identifies certain viral strains from the samples obtained from individuals with suspected COVID-19.
The new images of COVID-19 obtained by electron microscopy use electrons and magnets to focus and produce images, rather than using a conventional microscope based on the application of light and glass lenses.
The shape of the virus can be determined by electron microscopy and is used to classify the virus type. The scanning electron microscopy images produce a 3-D structure of the COVID-19 viruses, which shows the nucleocapsid protein (N-protein), and the spike protein, which appears to be blurry on the outside of the coat.
The two main types of electron microscopes produce different images. Transmission electron microscopes produce a flat image, and scanning electron microscopes create 3D-like pictures. All the images from all types of electron microscopes are black and white. The images obtained of the coronavirus have been artificially coloured. These images were obtained by the transmission electron microscope, which was isolated from the first U.S. case of COVID-19.
The Diversity of Electron Microscopes: A Comparison of Techniques and Uses
Disclaimer
The content of this article is intended for informational and educational purposes only. While every effort has been made to ensure accuracy, Open Medscience does not guarantee the completeness, reliability, or scientific validity of the information provided. The descriptions of electron microscopy techniques and their applications in virology reflect the state of knowledge at the time of publication (11 July 2021) and may not include the most current advancements or consensus.
This article does not provide medical advice, diagnostic guidance, or professional consultation. Readers should seek the advice of qualified healthcare or scientific professionals before acting on any information contained herein. The images referenced or described in the article, including depictions of the SARS-CoV-2 virus, may be artistically enhanced and should not be interpreted as exact scientific representations.
Open Medscience disclaims any liability for decisions made based on the information presented. External references and historical context are included for illustration and do not imply endorsement or current applicability.