Molecular imaging technology, such as PET, enables clinical microdose studies, providing vital insights into drug pharmacokinetics and biological processes.
A Glimpse into PET Imaging: Understanding the Basics
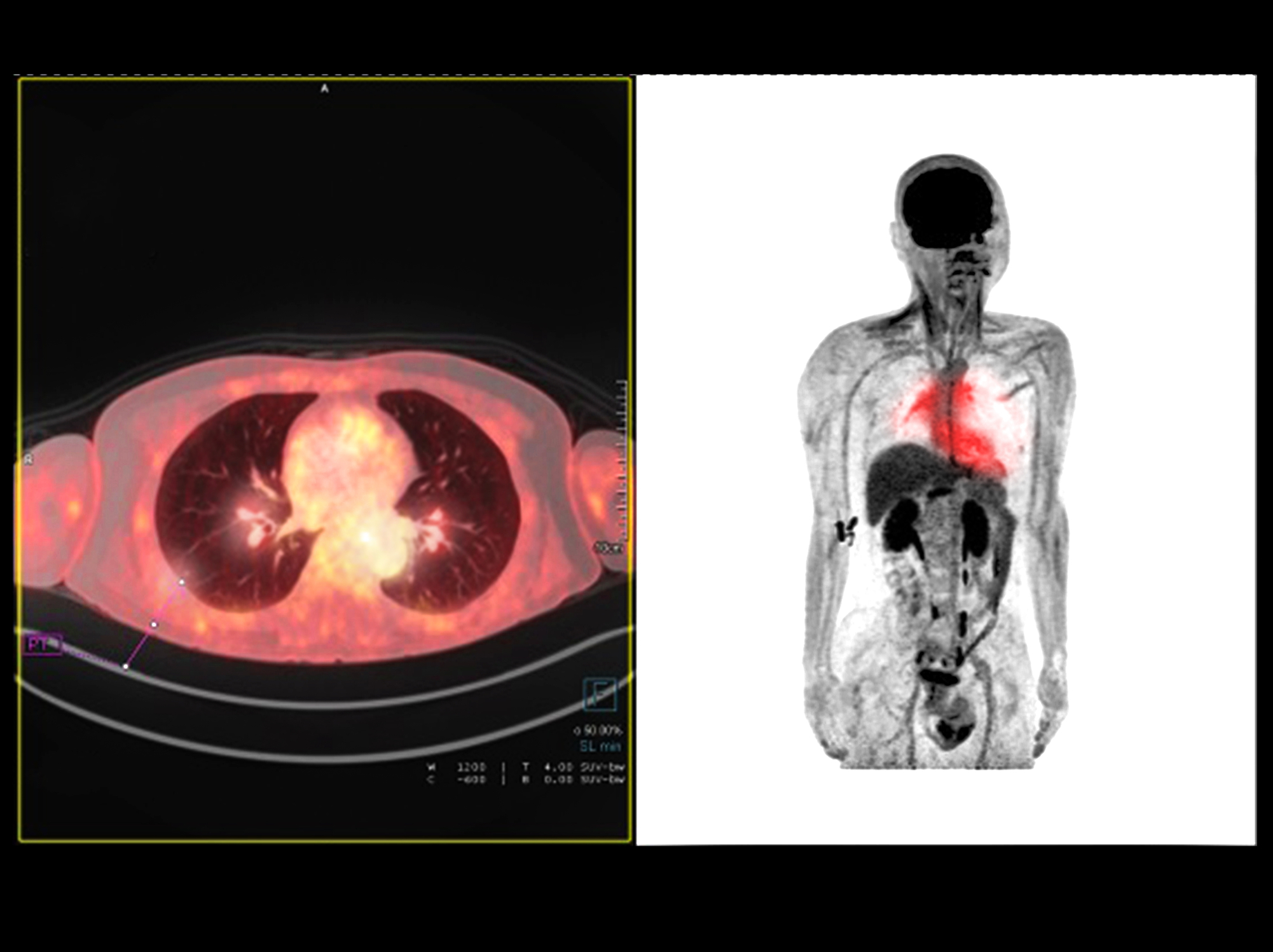
The powerful in vivo non-invasive imaging technique called positron emission tomography (PET) can generate dynamic information on biochemical processes in the human body. PET imaging is primarily used in oncology, neurology and cardiology. PET is an ultra-sensitive technique that can be used in human microdose studies.
A human Phase 0 microdose study is performed at the earliest stage of drug development. This approach enables preliminary data to be compiled on a human drug candidate before the commencement of Phase I clinical trials. PET imaging relies on detecting gamma radiation emitted from positrons during the annihilation of an electron.
PET Imaging in Humans: Exploring the Dual Roles of FDG and Drug Candidate Labeling
- The most common PET studies are performed with the radiotracer [18F]-fluorodeoxyglucose (FDG).
- The other type of PET study uses the labelling of a drug candidate.
PET studies in humans are carried out using a low dose of the drug (micrograms) administered intravenously. The microdose study aims to provide information about the rate at which the drug passes through the blood-brain barrier (disposition of the drug) and gain insight into human pharmacodynamics and pharmacokinetics. Advances in PET technology have significantly contributed to the drug discovery process by compiling more accurate data on drug efficacy and targeting the efficiency of potential lead compounds. The potential benefits of introducing PET imaging in the early phase of drug discovery will reduce the time spent on poor lead compounds as potential drug candidates. Also, PET imaging is used in late-stage drug discovery to investigate the toxicity and dosing studies, including tracking the response.
However, the main problem of PET imaging is the ability to synthesise be-spoke PET probes to investigate a particular disease. This is because of the challenge to develop feasible synthesis routes to incorporate the short-lived radiolabel to obtain probes in a reasonable radiochemical yield within the timescale associated with the short half-lives of PET radionuclides. Developing these synthesis methods is beneficial when using the shorter-lived isotopes (carbon-11, half-life = 20.3 minutes) in applying a broad range of targeting agents as probes. Automation will be vital in a novel probe containing fluorine-18 (half-life = 109.7 minutes) and carbon-11 PET isotopes. Enhancing the range of synthetic precursors labelled with PET isotopes also allows access to a broader range of specially designed molecular imaging agents with specific purposes, either to enhance specificity and sensitivity or to broaden the range of disease states that can be imaged.
Currently, 80% of clinical PET imaging and research uses FDG. However, the use of radiometals in PET imaging, particularly copper-64 (half-life = 12.7 hours), gallium-68 (half-life = 68 minutes) and yttrium-90 (half-life = 64.1 hours). Copper-64 and gallium-68 are routinely used in neuro and cardiac imaging. The advantage of using gallium-68 is that it does not require a cyclotron, therefore widening PET imaging opportunities throughout the world.
Why PET Imaging? Exploring the Rationale Behind this Diagnostic Tool
PET/CT offers a means of significantly reducing the drug discovery pipeline costs by providing the following:
- more secure identification of pre-clinical candidate
- robust means of securing a pass/fail decision in Phase I/First-in-Man trials because human bio-distribution, pharmacodynamics and pharmacokinetics can be measured.
The decision to move to Phase II is based on human data. PET/CT can provide an objective and quantitative endpoint for therapeutic efficacy in clinical trials. For example, investigating the reduction in tumour volume from FDG PET, proliferation using FLT (18F-fluorothymidine) and dopamine receptor activity using 11C-raclopride.
PET/CT could also reveal when drugs effectively target a selected population (a specific human phenotype) but fail when the appropriate subject is used. This opens the way for rational targeted drug design.
Pre-clinical PET/CT allows in vivo bio-distribution, pharmacodynamics, and pharmacokinetics to be measured. Receptor binding (K and Bmax) can be established in vivo. Objective and quantitative measurements of candidate therapeutics efficacy can be determined using known radiotracers. Given appropriate animal models, drugs targeting a specific genotype can be identified.
PET/CT can be used to discover why an established drug is active in one situation but not in a similar clinical situation, for example, gemcitabine for lung cancer compared to pancreatic cancer. Similarly, a drug’s complex mode of action, for example, carbamazepine, can be investigated in vivo both in animal models and in man.
PET Imaging in Drug Development: Answering Critical Early-Stage Questions
- Can a drug reach its intended target?
- Does it sufficiently bind the primary target?
- Does its interaction with the primary target affect one or more biochemical/signalling pathways?
- Is the interaction of the drug on cell or organ physiology relevant to a therapeutic effect?
- Does the drug interact with or affect other off-target tissues or organs?
- Can the drug be delivered to the target efficiently and achieve therapeutic levels?
The application and access to functional metabolic imaging have significantly enhanced the understanding of various pathological processes such as cancer. In addition, these imaging modalities have enabled the incorporation of information into patient management protocols. The leading molecular imaging tool is positron emission tomography (PET) which has emerged at the forefront of various clinical applications.
The clinical use of PET was restricted until the commercial use of medical cyclotrons in the 1980s in the field of neurology and cardiology. These cyclotron advancements made it practical to produce short-lived PET radiotracers at the hospital nuclear medicine departments. However, in the 1990s, the clinical applications of PET in oncology started with the radiotracer 18F-fluorodeoxyglucose (FDG) and, more recently, the hybrid of computed tomography (CT) fused with PET. These hybrid scanners combine the anatomic images produced by CT with the functional information of PET.
The principle of PET imaging is the ability to detect changes in cells, organs and tissues of the human body during metabolic processes. Some of these target glucose metabolism, enabling PET imaging to quantify functions within the cell. For example, in cancer cells, metabolic changes occur before the cells undergo certain changes, such as metaplasia. In the late stages of cancer, PET will follow the structural changes and detect changes in the disease state. The anatomical imaging techniques include CT and MRI, identifying any conditions in the organs.
Positron-Emitting Radioisotopes: The Stars of PET Imaging
The radiotracer 18F-fluorodeoxyglucose (FDG) behaves as an analogue of glucose and is used in glucose metabolism. FDG-PET radiotracer enters the cells like glucose, which involves phosphorylation by hexokinase to produce FDG-6-phosphate (FDG-6-P). The FDG-6-P does not undergo further metabolism but accumulates in the cells. This is because malignant cells have a higher rate of glycolysis than healthy cells.
The PET radiotracers 11C-thymidine and 18F-fluorothymidine (FLT) are both analogues of thymidine and are used as probes for cellular proliferation. Subsequently, the uptake of FLT has a similar mechanism to FDG uptake. The FLT uptake by the proliferation of cells does not incorporate into the DNA and only accumulates in intracellular tumour cells. FLT also can predict the tumour grade in lung cancer and is even a predictor of the brain tumour response. Furthermore,11C-methionine has been demonstrated in the evaluation of brain tumours. In addition, 11C-choline and 11C-acetate have been used in the PET imaging of prostate cancer to evaluate metastatic disease.
The PET radiotracer, rubidium-82, is used to evaluate the myocardial perfusion in a similar procedure when using technetium-99m (Myoview), thallium 201 or nitrogen-13 labelled ammonia.
PET imaging of the human skeleton can be investigated using the radiotracer 18F sodium fluoride and is comparable to technetium-99 labelled methylene diphosphonate.
Illuminating the Brain: PET Radiotracers and Their Role in Neuroimaging
PET Imaging in Medicine: Transforming Diagnosis and Treatment
Clinical Application Function
Brain tumours Gliomas are the most common of primary brain tumours in adults.
PET imaging studies have shown that the accumulation of FDG uptake is specific to the tumour type.
FDG-PET is used for tumour grading and response to the type of therapy.
Several PET radiotracers are used to investigate brain tumours and include 11C-methionine and the transport amino acid FLT. In addition to 11C-thymidine and 11C-choline used to study lipid metabolism.
Breast cancer In most cases, PET imaging is not used in the initial diagnosis and screening of patients with breast cancer.
However, PET can be used in the later staging of the disease, including restaging and detecting recurrent disease and therapy monitoring.
Cervical cancer FDG-PET can detect cervical cancers and can help in the personalised patient treatment plan.
Colorectal cancer PET imaging can detect primary colon carcinoma in at least 90% of cases compared to 60% sensitivity of computed tomography (CT).
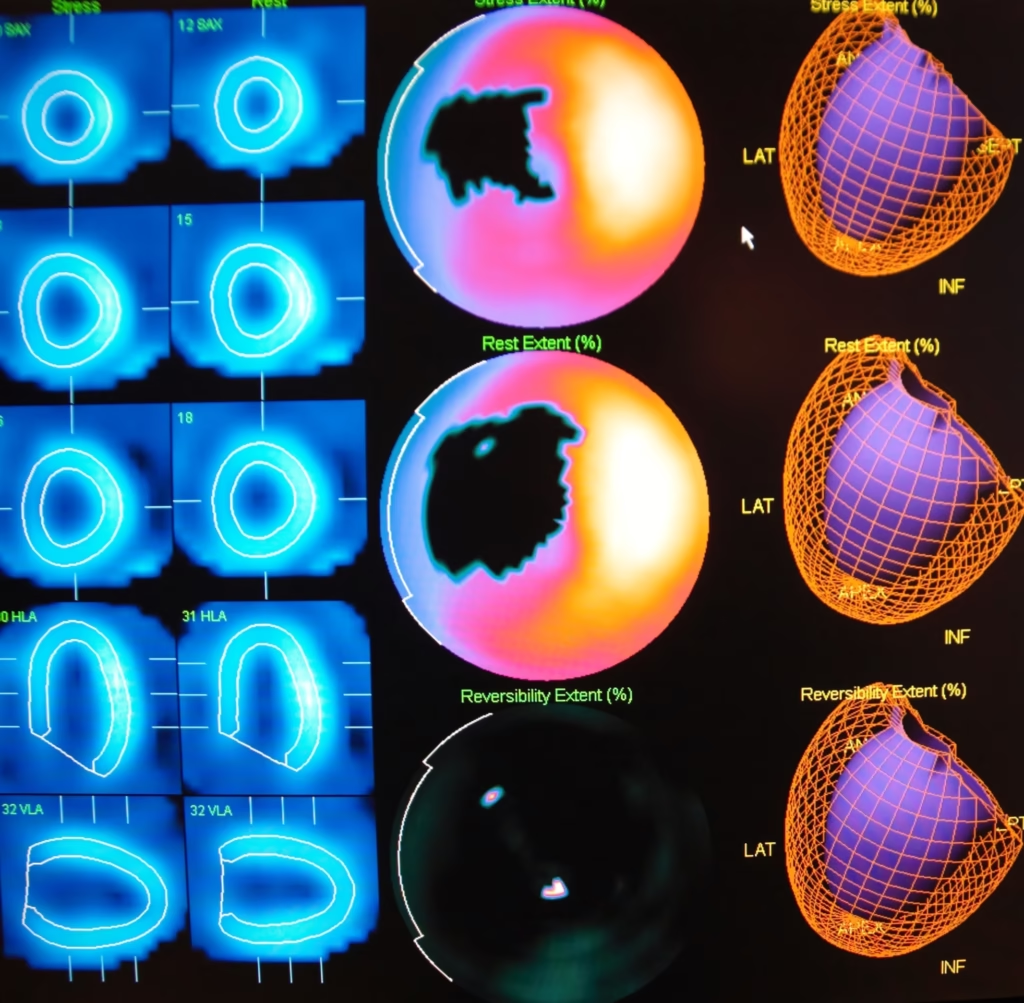
Coronary artery PET-MPI (myocardial perfusion imaging) performed during the stress and rest state of a patient. Radiotracers used in these tests are rubidium-82 or 13N-ammonia.
Coronary perfusion An absolute myocardial blood flow test used to evaluate the coronary artery lesion and assess the overall coronary vasculature.
Dementia PET imaging can help to diagnose the type of dementia.
FDG-PET shows hypometabolism in superior parieto-temporal cortices in Alzheimer’s disease.
PET can show dementia with Lewy bodies involvement of occipital lobes.
Epilepsy Ictal PET imaging can show increased glucose uptake at the site of seizure.
Head & Neck tumours PET/CT imaging can assist medical physicists in planning the radiation treatment for head and neck cancers.
The most prevalent cancer of the head and neck is squamous cell carcinoma.
PET can be used to find a primary tumour in patients with metastatic cervical lymphadenopathy.
PET is used in follow up for detection of recurrent disease and any changes in the tumour.
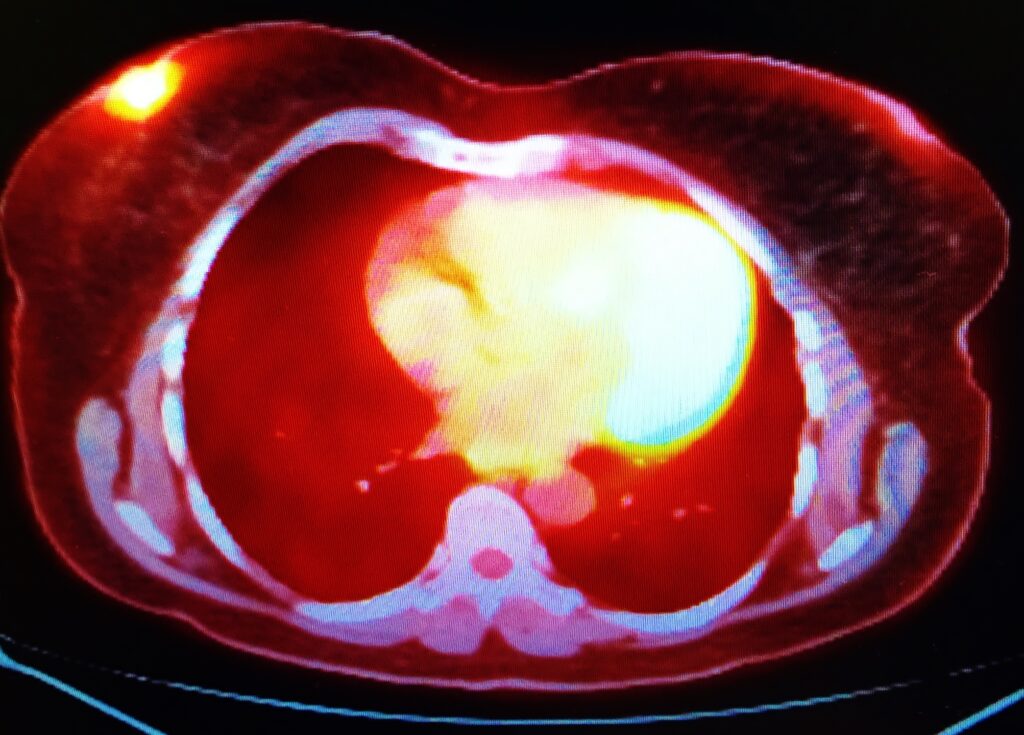
Lung cancer FDG-PET can be used to differentiate benign from malignant nodules in solitary pulmonary nodule (SPN).
Several lung cancers accumulate FDG except for pulmonary carcinoid and bronchioloalveolar carcinoma (BAC).
PET imaging is used in the determination of the nodal stage of non-small cell lung carcinoma (NSCLC).
In most cases, the staging of lung cancer is based on CT imaging.
FDG-PET scanning can identify positive nodes which are smaller than 10 mm in size.
FDG uptake correlates tumour proliferation in lung cancers.
FDG-PET can be used to detect metastases.
Lymphoma Lymphomas are classified as Hodgkin’s and non-Hodgkin’s Lymphoma (NHL).
PET imaging is a useful modality for staging, monitoring therapy and restaging in lymphomas.
PET is used in the evaluation of the spleen and bone marrow.
Melanoma The survival from melanoma depends on the stage at the time of diagnosis and thickness of the primary lesion.
PET is useful in the detection of metastases.
Movement disorders PET can image the dopaminergic receptor and 18F-fluoro DOPA scan is used to investigate Parkinson’s disease.
Musculoskeletal tumours Malignant primary bone tumours can be detected by PET imaging.
Also, differentiate the giant cell tumour, eosinophilic granuloma and fibrous dysplasia in benign environments.
Myocardial perfusion FDG-PET perfusion imaging is used to identify regions of the myocardium by comparing the regional FDG uptake with regional blood flow.
Oesophagal cancer Squamous cell carcinoma is prevalent in the upper part of the oesophagus.
The lower part of the oesophagus usually finds adenocarcinoma.
The overall sensitivity of PET-CT in carcinoma oesophagus is 95%.
Oncology PET and the hybrid PET-CT are both used in the staging of cancer.
FDG is the most used radiotracer in oncology.
PET imaging aims to detect any remaining lesions and the type of lesion. Also, used in the development of a personalised treatment plan.
PET-CT hybrid imaging is used to obtain biopsies from the active region of the tumour.
Renal, bladder and prostate cancers PET is limited in the diagnosis of primary tumours of the prostate, bladder and kidneys because of low uptake of FDG by the tumours.
The radiotracers 11C-choline 11C-acetate and been used to detect a primary lesion in prostatic cancers.
Stroke MRI and CT imaging can be used in the diagnosis of a stroke.
Testicular cancer Both seminoma and non-seminomatous germ cell tumours are found in the retroperitoneal nodes.
Conclusion
Over the last decade, PET has emerged as an important molecular imaging tool in the area of oncology, cardiology and neurology. The advancement of PET technology will produce high-resolution PET scanners with integrated multiple-row detectors of computed tomography. These PET-CT hybrid scanner aims to produce a scan under 5-min with reduced radiation that affects both the patient and radiographer. In addition, these new scanners will enhance patient comfort and minimise the effects of patient movement and increase patient throughput. The design of specific radiotracers by using automated synthesis will enable PET and PET/CT to facilitate more personalised imaging towards the patient’s treatment plan.
Disclaimer
The content provided in this article is intended for informational and educational purposes only and does not constitute medical, scientific, or professional advice. Open MedScience does not endorse any specific technologies, treatments, or protocols discussed herein. While every effort has been made to ensure accuracy at the time of publication, readers are advised that advances in the field may render some information outdated or subject to revision.
The use of positron emission tomography (PET) in clinical microdose studies is a specialised area of medical imaging research. Any decisions regarding the application of PET in clinical practice or drug development should be made in consultation with qualified healthcare professionals, researchers, or regulatory bodies, and in line with current guidelines and ethical approvals.
Neither Open MedScience nor the authors assume liability for any loss or harm resulting from the use or interpretation of the information presented in this article.
home » blog » nuclear medicine imaging »