The diagnostic breast imaging tool Positron Emission Mammography uses short-lived positron isotopes to detect breast cancer.
Positron Emission Mammography Imaging – PEM
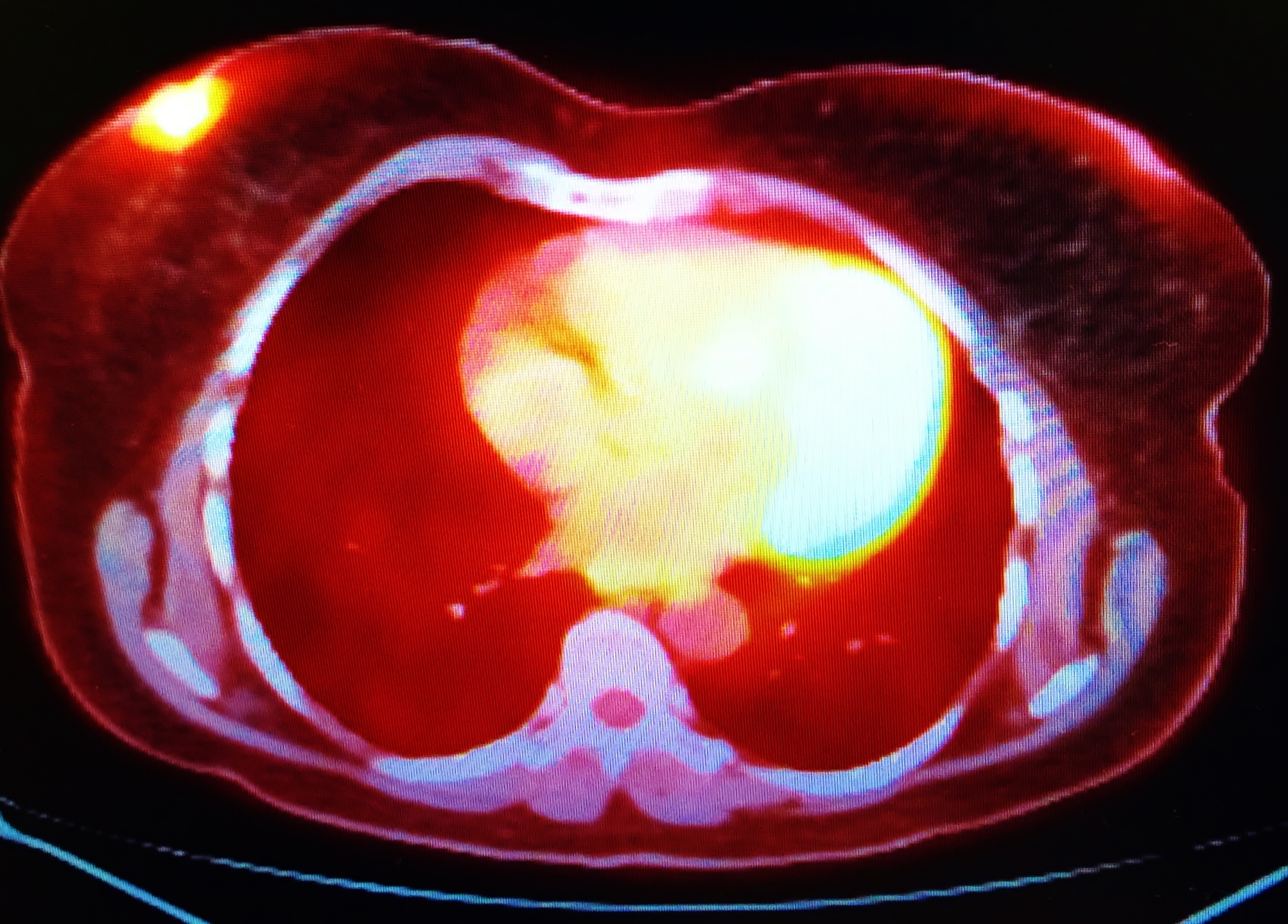
The diagnostic breast imaging tool Positron Emission Mammography (PEM) uses short-lived positron-emitting isotopes to generate high-resolution tomographic images of cancer within the breast.
PEM works by using an intravenous injection of the radiopharmaceutical 2-deoxy-2-(18F)fluoro-D-glucose, abbreviated as 18F-FDG. Therefore, the radiopharmaceutical is based on the design of the radioisotope fluorine-18 (half-life = 109.8 mins), which is attached to the delivery compound deoxyglucose to produce 18F-FDG. The cancer cells then absorb This modified radiolabelled glucose via the glucose transporter 1 system. The fundamental principle of PEM technology works on the premise that cancer cells display a high uptake of glucose. This imaging agent is effective in patients with dense breast tissue that may present with multiple lesions.
The radiopharmaceutical (18F-FDG) enters the cancer cell undergoing phosphorylation and cannot be transported back out of the cell. This process then leads to the accumulation of the imaging agent, and because the fluorine-18 nucleus is unstable, it undergoes a decay process, continually emitting positrons. The positron collides with an electron in the tumour tissue, which results in annihilation and produces two 511 keV gamma rays emitted in opposite directions. During the PEM scan, the gamma rays are detected when they strike a pair of detectors that are placed between the breasts. The detected gamma rays are then amplified by a photon-sensitive photomultiplier, which translates into an electrical signal that becomes an image.
PEM breast imaging
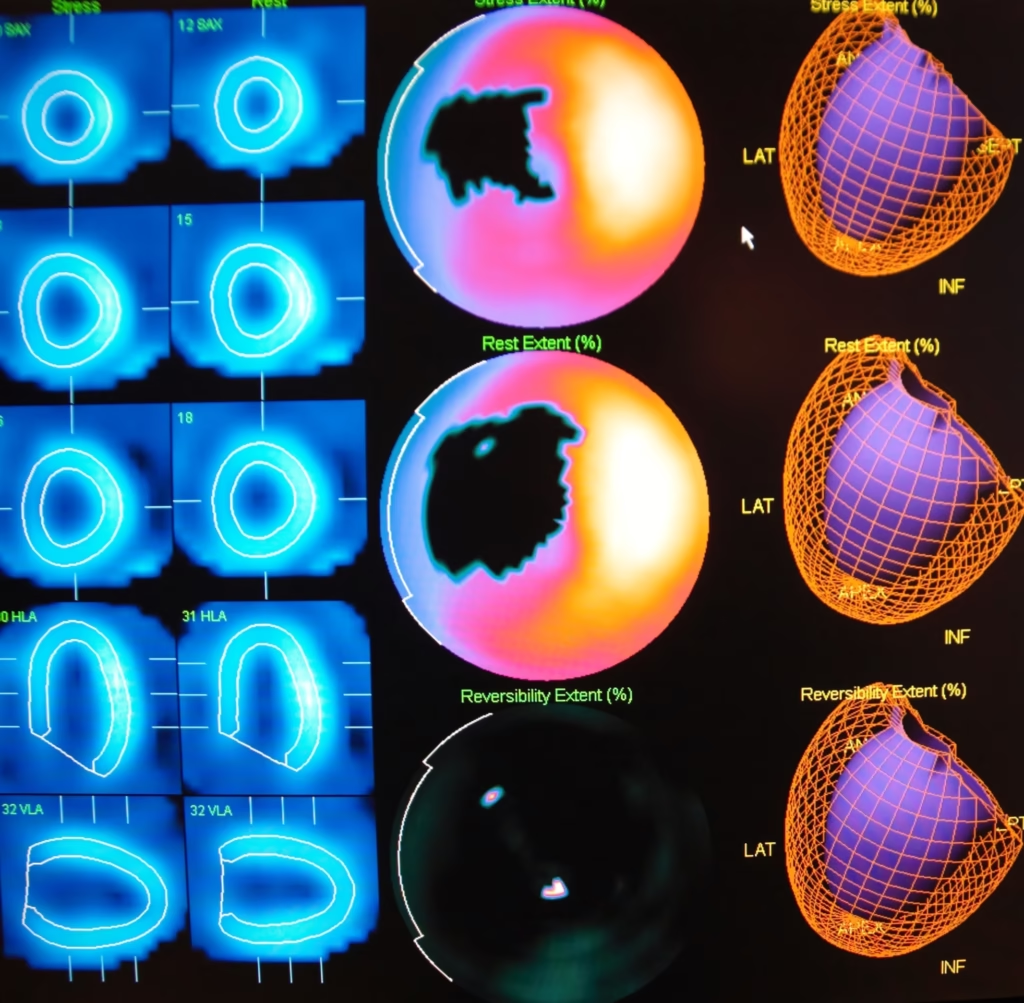
Positron Emission Mammography is currently part of the diagnostic toolkit to help assess patients who have detectable abnormalities in their mammogram. Both PEM and PET (positron emission tomography) are able to provide functional imaging by using the radiotracer 18F-FDG. However, PEM is primarily used for small body parts and utilises gentle immobilisation of the breast to attain higher spatial resolution: 1-2 mm for PEM and 4-6 mm for PET. The crystal detectors in PEM are constructed to provide this improved spatial resolution, including 1.5 mm in-plane and 5 mm between planes. Also, the combination of PET and CT (computed tomography) scanners using 18F-FDG is beneficial for the staging and restaging of advanced breast carcinoma.
In addition, PEM was approved by the US Food and Drug Administration and has been introduced into the clinical setting as a diagnostic aid to mammography and breast ultrasonography. Furthermore, PEM is currently under clinical investigation to improve the sensitivity of breast cancer screening programmes. The indications for PEM include:
- the initial staging evaluation of patients with diagnosed cancer;
- distinguishing recurrent carcinoma from scar tissue;
- monitoring response to chemotherapy treatment
PEM has high imaging sensitivity for breast lesions. However, its clinical utility requires further investigation. Nevertheless, PEM cannot provide the anatomical detail that is provided by magnetic resonance imaging (MRI).
Positron Emission Mammography (PEM) is a specialised diagnostic breast imaging tool that uses short-lived positron-emitting isotopes to generate high-resolution tomography images of cancer within the breast. This advanced imaging technique offers a unique approach to detecting and evaluating breast cancer, especially in patients with dense breast tissue or multiple lesions.
Mechanism of PEM Imaging
PEM operates by administering an intravenous injection of the radiopharmaceutical 2-deoxy-2-(18F)fluoro-D-glucose (18F-FDG). This compound consists of the radioisotope fluorine-18, which has a half-life of approximately 109.8 minutes, attached to deoxyglucose. The modified radiolabelled glucose is absorbed by cancer cells via the glucose transporter 1 system, exploiting the principle that cancer cells exhibit a high uptake of glucose.
Once inside the cancer cell, 18F-FDG undergoes phosphorylation and becomes trapped, accumulating the imaging agent. The unstable fluorine-18 nucleus decays, emitting positrons that collide with electrons in the tumour tissue. This collision results in annihilation, producing two 511 keV gamma rays emitted in opposite directions. During a PEM scan, these gamma rays are detected by a pair of detectors placed between the breasts. The detected gamma rays are then amplified by photon-sensitive photomultipliers, translating them into electrical signals processed into high-resolution images.
Clinical Applications of PEM
PEM is part of the diagnostic toolkit used to assess patients with detectable abnormalities on their mammograms. While both PEM and positron emission tomography (PET) use the radiotracer 18F-FDG to provide functional imaging, PEM is specifically designed for imaging small body parts like the breast. It employs gentle immobilisation of the breast to achieve higher spatial resolution: 1-2 mm for PEM compared to 4-6 mm for PET. The crystal detectors in PEM provide enhanced spatial resolution, including 1.5 mm in-plane and 5 mm between planes. Additionally, PET/CT scanners using 18F-FDG are beneficial for staging and restaging advanced breast carcinoma.
PEM has been approved by the US Food and Drug Administration and is utilised in clinical settings as a diagnostic aid alongside mammography and breast ultrasonography. It is under clinical investigation to improve the sensitivity of breast cancer screening programmes. Indications for PEM include:
- Initial staging evaluation of patients with diagnosed cancer
- Distinguishing recurrent carcinoma from scar tissue
- Monitoring response to chemotherapy treatment
Advantages and Limitations
PEM offers high imaging sensitivity for breast lesions, making it a valuable tool in detecting and evaluating breast cancer. Its ability to provide functional imaging of glucose metabolism in cancer cells adds a significant dimension to breast cancer diagnosis and management. However, PEM does not provide the anatomical detail that magnetic resonance imaging (MRI) offers. While PEM is effective in highlighting metabolic activity and detecting cancerous lesions, MRI is superior in visualising anatomical structures and providing detailed images of breast tissue.
Future Directions
Ongoing clinical investigations aim to enhance the sensitivity and specificity of PEM, potentially integrating it more robustly into breast cancer screening protocols. Researchers are exploring ways to combine PEM with other imaging modalities to provide a comprehensive assessment of breast cancer, improving diagnostic accuracy and patient outcomes. The continued development and refinement of PEM technology hold promise for its expanded use in clinical practice, offering a powerful tool in the fight against breast cancer.
In conclusion, Positron Emission Mammography (PEM) represents a significant advancement in breast imaging. It provides high-resolution, functional images that aid in diagnosing and managing breast cancer. Its unique capabilities complement other imaging modalities, offering a comprehensive breast cancer detection and treatment monitoring approach.
Disclaimer
The content presented in this article is intended for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Positron Emission Mammography (PEM) is a specialised imaging technique that may not be suitable for all patients, and its use should be determined by qualified healthcare professionals based on individual clinical circumstances. The information reflects knowledge available as of the publication date (26 April 2018) and may not include the most recent research developments or clinical guidelines. Readers are advised to consult their doctor or a medical specialist for personalised advice or questions regarding any medical condition. Open Medscience does not accept any responsibility for the outcomes of medical decisions based on the use or interpretation of information contained in this article.
home » blog » nuclear medicine imaging »